Amplite® Fluorimetric Caspase 3/7 Assay Kit *Green Fluorescence*
| Price | |
| Catalog Number | |
| Unit Size | |
| Quantity |
| Telephone | 1-800-990-8053 |
| Fax | 1-800-609-2943 |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Shipping | Standard overnight for United States, inquire for international |
| Extinction coefficient (cm -1 M -1) | 80000 |
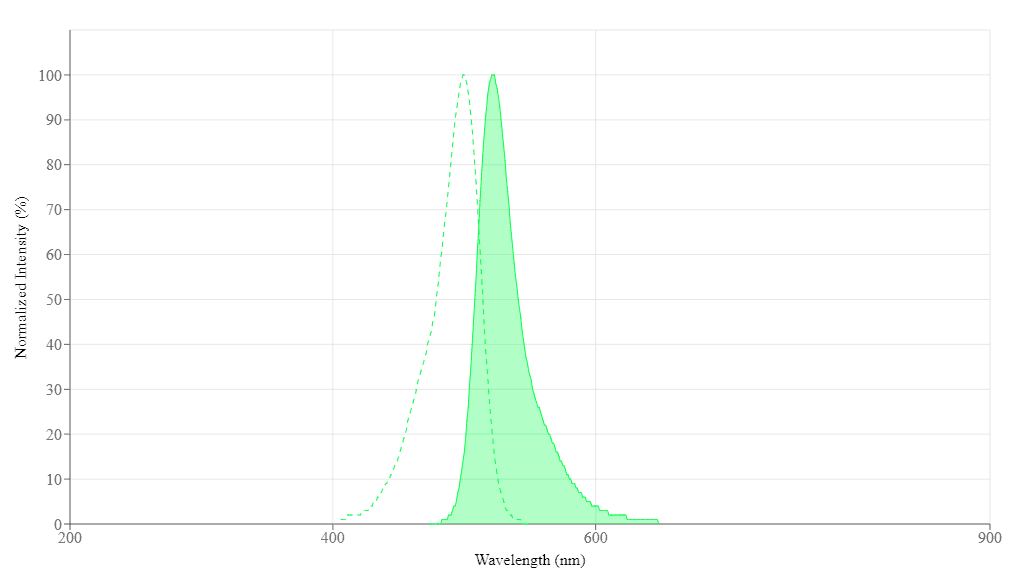
| Excitation (nm) | 500 |
| Emission (nm) | 522 |
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
| Overview |
Extinction coefficient (cm -1 M -1) 80000 | Excitation (nm) 500 | Emission (nm) 522 |
Platform
Fluorescence microplate reader
| Excitation | 490 nm |
| Emission | 525 nm |
| Cutoff | 515 nm |
| Recommended plate | Solid black |
Components
Example protocol
AT A GLANCE
Protocol summary
- Prepare cells with test compounds (100 µL/well/96-well plate or 25 µL/well/384-well plate)
- Add equal volume of Caspase 3/7 working solution (100 µL/well/96-well plate or 25 µL/well/384-well plate)
- Incubate at room temperature for 1 hour
- Monitor fluorescence intensity at Ex/Em = 490/525 nm (Cutoff = 515 nm)
Important notes
Thaw component A, B, C (if desired, Component D and E) at room temperature before starting the experiment.
PREPARATION OF STOCK SOLUTION
1. Caspase 3/7 Inhibitor Ac-DEVD-CHO stock solution (1 mM):
Add 100 µL of DMSO (not provided) directly to the vial of Ac-DEVD-CHO (Component D), and mix well to make 1 mM Caspase 3/7 Inhibitor Ac-DEVD-CHO stock solution. This Caspase 3/7 Inhibitor can be used to confirm the correlation between fluorescence signal intensity and Caspase 3/7-like protease activities.
PREPARATION OF WORKING SOLUTION
Add 50 μL of 200X Caspase 3/7 Substrate stock solution (Component A) and 100 μL of 1M DTT solution (Component C) into 10 mL of Assay Buffer (Component B) and mix well to make Caspase 3/7 working solution. Note: This Caspase 3/7 working solution is enough for 100 assays using a reaction volume of 100 μL per assay. Protect from light.
For guidelines on cell sample preparation, please visit
https://www.aatbio.com/resources/guides/cell-sample-preparation.html
SAMPLE EXPERIMENTAL PROTOCOL
- Treat cells with 10 µL of 10X test compound (for a 96-well plate) or 5 µL of 5X test compound (for a 384-well plate) in PBS or desired buffer. For blank wells (medium without the cells), add the corresponding amount of compound buffer.
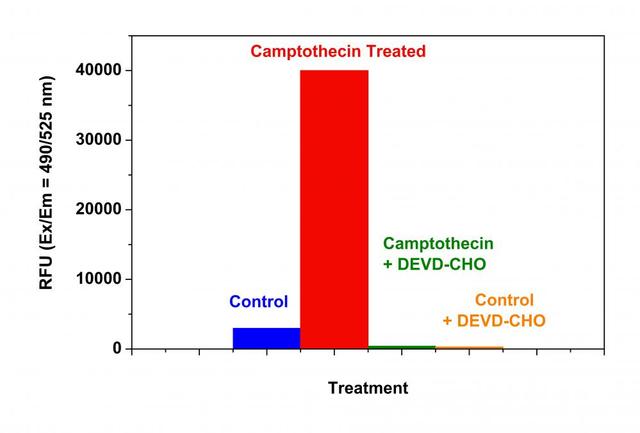
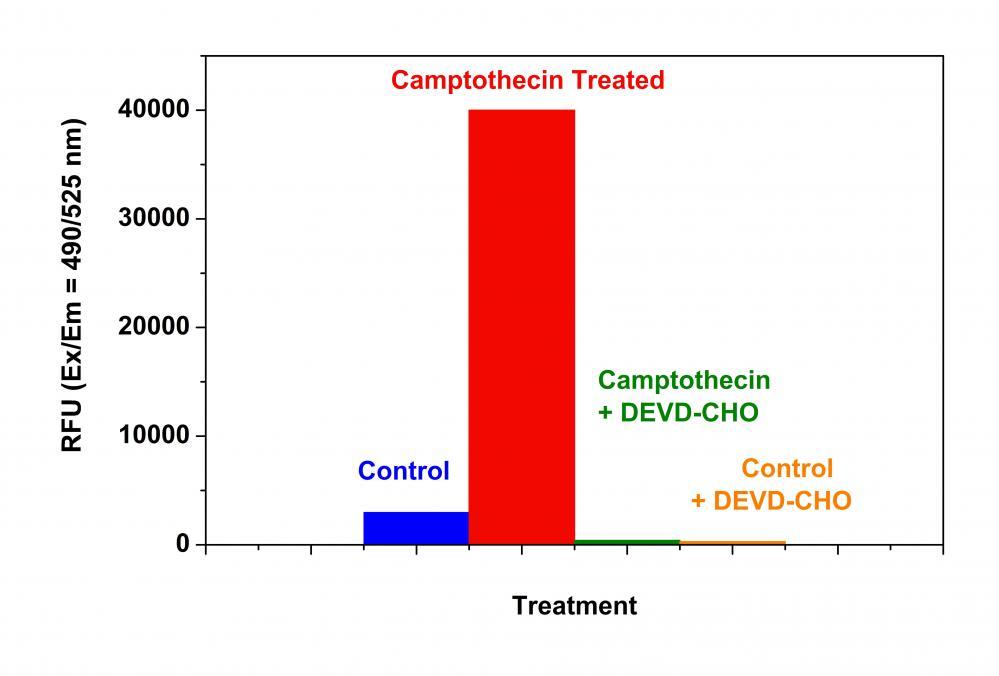
- Incubate the cell plates in an incubator for desired period of time ( 4 - 6 hours for Jurkat cells treated with camptothecin) to induce apoptosis.
- Add 100 µL/well (96-well plate) or 25 µL/well (384-well plate) of Caspase 3/7 working solution.
- Incubate the plate at room temperature for at least 1 hour, protected from light. Note: If desired, add 1 µL of 1 mM Caspase 3/7 Inhibitor Ac-DEVD-CHO stock solution into selected samples 10 minutes before adding Caspase 3/7 working solution at room temperature to confirm the Caspase 3/7-like activities. Note: If desired, prepare a R110 standards by diluting 5 mM R110 Standard (Component E) into growth Medium to yield serially diluted R110 standards ranging from 0 - 50 µM. Add 100 µL of the serially diluted R110 standards into the wells containing 100 µL of Caspase 3/7 working solution at any time prior to measuring the fluorescence. This standard curve could be used to determine the moles of product produced in the caspase 3/7 containing reactions.
- Centrifuge cell plates (especially for the non-adherent cells) at 800 rpm for 2 minutes with brake off.
- Monitor the fluorescence increase at Ex/Em = 490/525 nm (Cutoff = 515 nm).
Spectrum
Spectral properties
| Extinction coefficient (cm -1 M -1) | 80000 |
| Excitation (nm) | 500 |
| Emission (nm) | 522 |
Product Family
| Name | Excitation (nm) | Emission (nm) |
| Amplite® Fluorimetric Caspase 3/7 Assay Kit *Blue Fluorescence* | 341 | 441 |
| Amplite® Fluorimetric Caspase 3/7 Assay Kit *Red Fluorescence* | 532 | 619 |
Images
Citations
Authors: Takakura, Masatoshi and Mizutani, Ayano and Kudo, Mizuki and Ishikawa, Airi and Okamoto, Takuya and Fu, Tong Xuan and Kurimoto, Shin-ichiro and Koike, Yuka and Mishima, Kenji and Tanaka, Junichi and others,
Journal: Biological and Pharmaceutical Bulletin (2024): 138--144
Authors: Minafra, Anna Rita and Rafii, Puyan and Mossner, Sofie and Bazgir, Farhad and Floss, Doreen M and Moll, Jens M and Scheller, J{\"u}rgen
Journal: Journal of Biological Chemistry (2023): 104989
Authors: Ettich, Julia and Wittich, Christoph and Moll, Jens M and Behnke, Kristina and Floss, Doreen M and Reiners, Jens and Christmann, Andreas and Lang, Philipp A and Smits, Sander HJ and Kolmar, Harald and others,
Journal: The Journal of Biological Chemistry (2023)
Authors: Ettich, Julia and Wittich, Christoph and Moll, Jens M and Behnke, Kristina and Floss, Doreen M and Reiners, Jens and Christmann, Andreas and Lang, Philipp A and Smits, Sander HJ and Kolmar, Harald and others,
Journal: Journal of Biological Chemistry (2023): 105270
Authors: Salvati, Annamaria and Melone, Viola and Sellitto, Assunta and Rizzo, Francesca and Tarallo, Roberta and Nyman, Tuula A and Giurato, Giorgio and Nassa, Giovanni and Weisz, Alessandro
Journal: Breast Cancer Research (2022): 1--23
Authors: Onodera, Risako and Morioka, Shunsuke and Unida, Shinshu and Motoyama, Keiichi and Tahara, Kohei and Takeuchi, Hirofumi
Journal: European Journal of Pharmaceutical Sciences (2022): 106081
Authors: H{\"a}hnel, Viola and Brosig, Andreas-Michael and Ehrenschwender, Martin and Burkhardt, Ralph and Offner, Robert and Ahrens, Norbert
Journal: Transfusion (2021)
Authors: Kanne, Julian and Hussong, Michelle and Isensee, J{\"o}rg and Mu{\~n}oz-L{\'o}pez, {\'A}lvaro and Wolffgramm, Jan and He{\ss}, Felix and Grimm, Christina and Bessonov, Sergey and Meder, Lydia and Wang, Jie and others,
Journal: Cell Death \& Disease (2021): 1--15
Authors: Suzuki, Ryusuke and Fujiwara, Yukio and Saito, Mitsuru and Arakawa, Shoutaro and Shirakawa, Jun-ichi and Yamanaka, Mikihiro and Komohara, Yoshihiro and Marumo, Keishi and Nagai, Ryoji
Journal: Journal of bone and mineral research (2020): 1992--2003
Authors: Cherif, Hosni and Bisson, Daniel G and Mannarino, Matthew and Rabau, Oded and Ouellet, Jean A and Haglund, Lisbet
Journal: Elife (2020): e54693
References
Authors: Lerma-Diaz JM, Hern and ez-Flores G, Dominguez-Rodriguez JR, Ortiz-Lazareno PC, Gomez-Contreras P, Cervantes-Munguia R, Scott-Algara D, Aguilar-Lemarroy A, Jave-Suarez LF, Bravo-Cuellar A.
Journal: Immunol Lett (2006): 149
Authors: Wu X, Simone J, Hewgill D, Siegel R, Lipsky PE, He L.
Journal: Cytometry A (2006): 477
Authors: Sakaue M, Motoyama Y, Yamamoto K, Shiba T, Teshima T, Chiba K.
Journal: Biochem Biophys Res Commun (2006): 878
Authors: Miller TJ, Schneider RJ, Miller JA, Martin BP, Al-Ubaidi MR, Agarwal N, Dethloff LA, Philbert MA.
Journal: Neurotoxicology (2006): 44
Authors: Xiao XL, Peng J, Su Q, Xiang SL, Tang GH, Huang YS, Zhou XT.
Journal: Ai Zheng (2006): 1247
Authors: Jiang DJ, Jia SJ, Dai Z, Li YJ.
Journal: J Mol Cell Cardiol (2006): 529
Authors: Fennell M, Chan H, Wood A.
Journal: J Biomol Screen (2006): 296
Authors: Kume T, Taguchi R, Katsuki H, Akao M, Sugimoto H, Kaneko S, Akaike A.
Journal: Eur J Pharmacol (2006): 69
Authors: O'Brien MA, Daily WJ, Hesselberth PE, Moravec RA, Scurria MA, Klaubert DH, Bulleit RF, Wood KV.
Journal: J Biomol Screen (2005): 137
Authors: Brauns SC, Dealtry G, Milne P, Naude R, Van de Venter M.
Journal: Anticancer Res (2005): 4197
Application notes
Design of potent inhibitors of acetylcholinesterase using morin as the starting compound
Acetylcholinesterase Inhibitory Activity of Pigment Echinochrome A
Induction of Neurite Outgrowth in PC12 Cells
Induction of Neuritogenesis in PC12 Cells by a Pulsed Electromagnetic Field
FAQ
How should I reconstitute an NADPH standard?
Will Amplite® Fluorimetric NAD/NADH Ratio Assay Kit *Red Fluorescence* work with NADP/NADPH? Can this kit measure NADP+ and NADPH?
What is the concentration of calcium inside cells?
What assay kits measure NADP/NADPH from cell samples?