Amplite® Fluorimetric Renin Assay Kit *Red Fluorescence*
| Price | |
| Catalog Number | |
| Unit Size | |
| Quantity |
| Telephone | 1-800-990-8053 |
| Fax | 1-800-609-2943 |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Shipping | Standard overnight for United States, inquire for international |
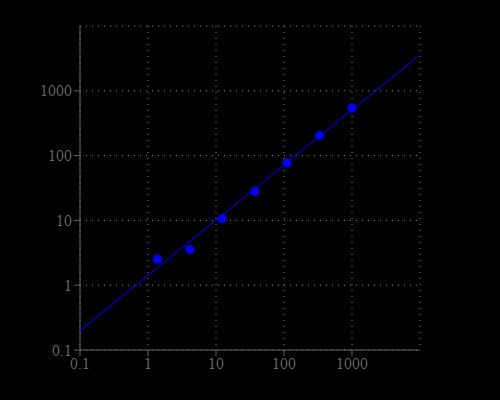
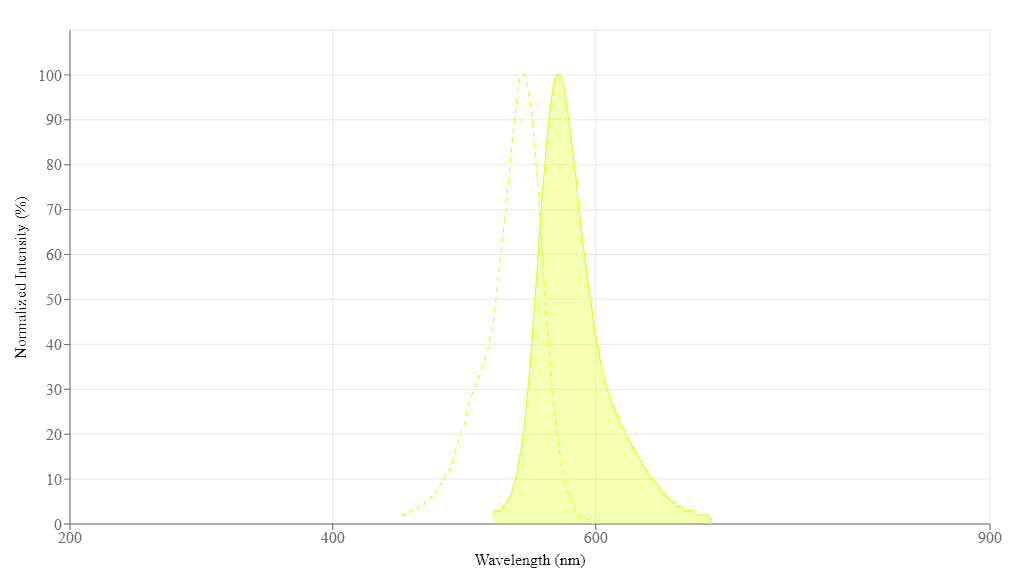
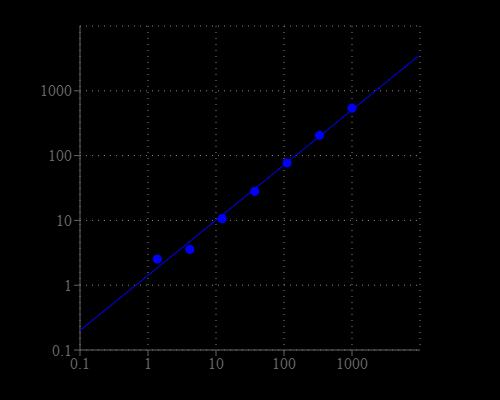
| Excitation (nm) | 545 |
| Emission (nm) | 572 |
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
| Overview |
Excitation (nm) 545 | Emission (nm) 572 |
Platform
Fluorescence microplate reader
| Excitation | 540 nm |
| Emission | 590 nm |
| Cutoff | 570 nm |
| Recommended plate | Solid black |
Components
Example protocol
AT A GLANCE
Prepare and add Renin standard and test samples (50 µL)
- Add Renin Red™ substrate working solution (50 µL)
- Incubate for 30 - 60 min at 37 °C incubator (for end point reading)
- Monitor fluorescence intensity at Ex/Em = 540/590 nm
Important Thaw all the kit components at room temperature before starting the experiment. Prepare Renin containing biological samples as desired.
PREPARATION OF STANDARD SOLUTIONS
https://www.aatbio.com/tools/serial-dilution/13530
PREPARATION OF WORKING SOLUTION
Add 50 μL of Renin Red™ Substrate (Component A) into 5 mL of Assay Buffer (Component C) to make a total volume of 5.05 mL.
Note The Renin Red™ Substrate should be used promptly.
SAMPLE EXPERIMENTAL PROTOCOL
Table 1. Layout of Renin standards and test samples in a solid black 96-well microplate. Ren = Renin Standards (Ren1-Ren7, 1 to 1000 ng/mL); BL = Blank Control; TS = Test Samples.
| BL | BL | TS | TS |
| Ren1 | Ren1 | ... | ... |
| Ren2 | Ren2 | ... | ... |
| Ren3 | Ren3 | ||
| Ren4 | Ren4 | ||
| Ren5 | Ren5 | ||
| Ren6 | Ren6 | ||
| Ren7 | Ren7 |
Table 2. Reagent composition for each well.
Note The Renin standards are for positive control only, and should not be relied on as a quantitation standard for enzyme activity.
| Well | Volume | Reagent |
| Ren1 - Ren7 | 50 µL | serial dilution (1 to 1000 ng/mL) |
| BL | 50 µL | Assay Buffer (Component C) |
| TS | 50 µL | sample |
- Prepare Renin containing biological samples as desired.
- Prepare the Renin standards and/or Renin-containing test samples according to the layout provided in Table 1 and Table 2. For a 384-well plate, use 25 µL of reagent per well instead of 50 µL.
- Pre-incubate the plate at a desired temperature for the enzyme reaction (e.g. 25 °C or 37 °C) for 10 - 15 min, if you are screening Renin inhibitors.
Add 50 µL (96-well) or 25 µL (384-well) of Renin Red™ substrate working solution to the Standard sample, control and test wells of the assay plate.
- Incubate the reaction at 37 °C incubator for 30 to 60 minutes.
Monitor the fluorescence intensity with a fluorescence plate reader at Ex/Em = 540/590 nm (cut off = 570 mn).
Note The selectivity of the renin substrate used in the kit has not been thoroughly tested. It may also respond to other proteases since peptide-based protease substrates generally have low selectivity. One might use a renin-specific inhibitor for its specific test, such as in the presence of a renin-specific inhibitor, hydrolysis of the substrate is only due to the non-specific protease activity. The difference between the total activity and the activity in the presence of renin specific Inhibitor gives the renin activity in the sample.
For kinetic reading: Immediately start measuring fluorescence intensity and continuously record data every 5 minutes for 30 to 60 minutes.
For end-point reading: Incubate the reaction at 37 °C for 60 minutes or longer, kept from light if possible. And then measure the fluorescence intensity.
Images
Citations
Authors: Akentieva, Natalia and Sanina, Natalia and Gizatullin, Artur and Shkondina, Natalia and Andreeva, Anna and Shram, Stanislav and Aldoshin, Sergei
Journal: (2022)
References
Authors: Vargas SL, Toma I, Kang JJ, Meer EJ, Peti-Peterdi J.
Journal: J Am Soc Nephrol (2009): 1002
Authors: Friis UG, Madsen K, Svenningsen P, Hansen PB, Gulaveerasingam A, Jorgensen F, Aalkjaer C, Skott O, Jensen BL.
Journal: J Am Soc Nephrol (2009): 2154
Authors: Wanka H, Kessler N, Ellmer J, Endlich N, Peters BS, Clausmeyer S, Peters J.
Journal: J Cell Mol Med (2009): 2926
Authors: Schmiedt CW, Hurley KA, Tong X, Rakhmanova VA, Po CL, Hurley DJ.
Journal: Am J Vet Res (2009): 1315
Authors: De Mello WC, Gerena Y.
Journal: Regul Pept (2009): 139
Authors: Imanishi T, Tsujioka H, Ikejima H, Kuroi A, Takarada S, Kitabata H, Tanimoto T, Muragaki Y, Mochizuki S, Goto M, Yoshida K, Akasaka T.
Journal: Hypertension (2008): 563
Authors: Du D, Kato T, Nabi AH, Suzuki F, Park EY.
Journal: Biotechnol Appl Biochem (2008): 195
Authors: Feldt S, Batenburg WW, Mazak I, Maschke U, Wellner M, Kvakan H, Dechend R, Fiebeler A, Burckle C, Contrepas A, Jan Danser AH, Bader M, Nguyen G, Luft FC, Muller DN.
Journal: Hypertension (2008): 682
Authors: Hanner F, von Maltzahn J, Maxeiner S, Toma I, Sipos A, Kruger O, Willecke K, Peti-Peterdi J.
Journal: Am J Physiol Regul Integr Comp Physiol (2008): R371
Authors: Glenn ST, Jones CA, Pan L, Gross KW.
Journal: Physiol Genomics (2008): 243
Application notes
Design of potent inhibitors of acetylcholinesterase using morin as the starting compound
Acetylcholinesterase Inhibitory Activity of Pigment Echinochrome A
Induction of Neurite Outgrowth in PC12 Cells
Induction of Neuritogenesis in PC12 Cells by a Pulsed Electromagnetic Field
FAQ
How should I reconstitute an NADPH standard?
Will Amplite® Fluorimetric NAD/NADH Ratio Assay Kit *Red Fluorescence* work with NADP/NADPH? Can this kit measure NADP+ and NADPH?
What is the concentration of calcium inside cells?
What assay kits measure NADP/NADPH from cell samples?