Indo-1, AM
CAS 112926-02-0
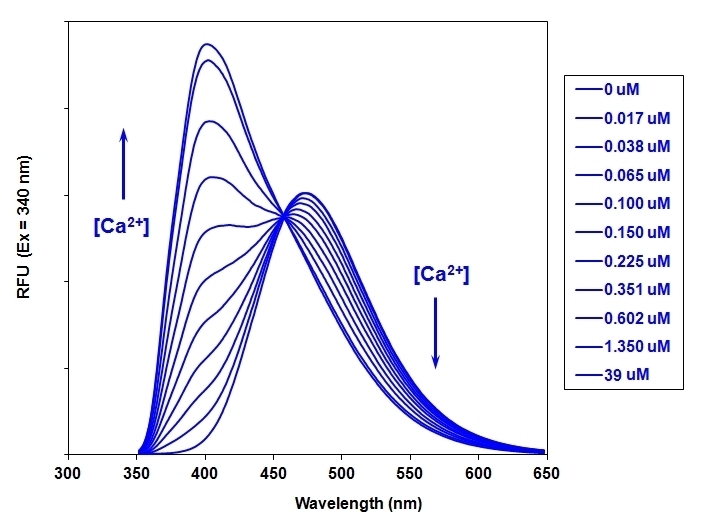
Indo-1 AM is a UV-excitable, cell-permeable ratiometric calcium indicator that shifts emission upon calcium binding, enabling quantitative measurement of intracellular calcium-ion dynamics.
- Ratiometric detection: Distinct shift in emission from 480 nm to 400 nm upon binding to calcium
- Optimized for flow cytometry: Requires only a single UV excitation source, such as 351–364 nm spectral lines, simplifying experimental setups
- Versatile applications: Compatible with various techniques, including fluorescence microscopy, spectroscopy, microplate readers, and flow cytometry

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 21030 | 1 mg | Price | |
| 21033 | 50 mg | Price |
Physical properties
| Dissociation constant (Kd, nM) | 230 |
| Molecular weight | 1009.91 |
| Solvent | DMSO |
Spectral properties
| Extinction coefficient (cm -1 M -1) | 33000 |
| Excitation (nm) | 330 |
| Emission (nm) | 404 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
| CAS | 112926-02-0 |
Instrument settings
| Fluorescence microscope | |
| Excitation | Indo-1 filter set |
| Emission | Indo-1 filter set |
| Recommended plate | Black wall/clear bottom |
| Fluorescence microplate reader | |
| Excitation | 340 |
| Emission | 400, 475 |
| Cutoff | Ex/Em = 340/400, no cut off. Ex/Em = 340/475, cut off 455 |
| Recommended plate | Black wall/clear bottom |
| Instrument specification(s) | Bottom read mode/Programmable liquid handling |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on September 20, 2024

