Calbryte™ 520 AM
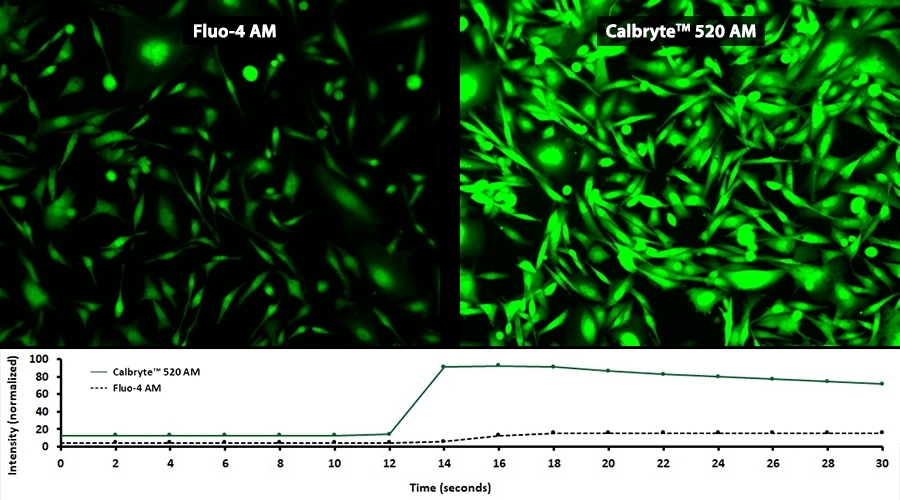
Calbryte™ 520 AM is a green-fluorescent calcium indicator with superior brightness and photostability that enables sensitive detection of calcium flux in live cells for GPCR screening and calcium signaling studies.
- High-quality Reagent: Manufactured to the highest standards for consistent results
- Reliable Performance: Proven effectiveness across multiple experimental conditions
- Versatile Applications: Suitable for a wide range of research and diagnostic applications

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 20650 | 2x50 ug | Price | |
| 20651 | 10x50 ug | Price | |
| 20653 | 1 mg | Price |
Physical properties
| Dissociation constant (Kd, nM) | 1200 |
| Molecular weight | 1090.90 |
| Solvent | DMSO |
Spectral properties
| Excitation (nm) | 493 |
| Emission (nm) | 515 |
| Quantum yield | 0.75 1 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
Instrument settings
| Flow cytometer | |
| Excitation | 488 nm laser |
| Emission | 530/30 nm filter |
| Instrument specification(s) | FITC channel |
| Fluorescence microscope | |
| Excitation | FITC |
| Emission | FITC |
| Recommended plate | Black wall/clear bottom |
| Fluorescence microplate reader | |
| Excitation | 490 |
| Emission | 525 |
| Cutoff | 515 |
| Recommended plate | Black wall/clear bottom |
| Instrument specification(s) | Bottom read mode/Programmable liquid handling |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 31, 2026

