Cell Meter™ Cell Viability Assay Kit
NIR Fluorescence Optimized for Fluorescence Microplate Reader
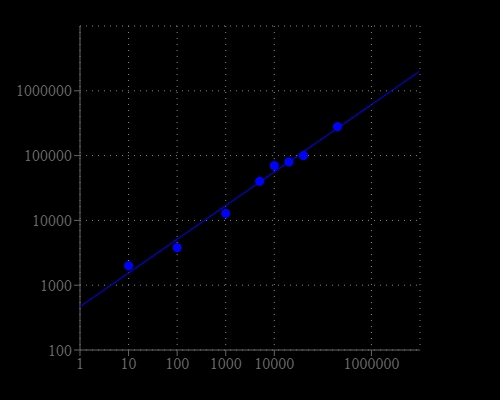
Our Cell Meter™ assay kits are a set of tools for monitoring cell viability. There are a variety of parameters that can be used for monitoring cell viability. This kit uses a proprietary dye that gets enhanced fluorescence upon entering into live cells. The dye is a hydrophobic compound that easily permeates intact live cells. The hydrolysis of the weakly fluorescent substrate by intracellular esterases generates a strongly fluorescent hydrophilic product that is well-retained in the cell cytoplasm. The esterase activity is proportional to the number of viable cells, and thus directly related to the fluorescence intensity of the product generated from the esterase-catalyzed hydrolysis of the fluorogenic substrate. Cells grown in black-walled plates can be stained and quantified in less than two hours. The assay is more robust than the tetrazolium salt or Alarmar Blue™-based assays. It can be readily adapted for high-throughput assays in a wide variety of fluorescence platforms such as microplate assays, immunocytochemistry and flow cytometry. It is useful for a variety of studies, including cell adhesion, chemotaxis, multidrug resistance, cell viability, apoptosis and cytotoxicity. The kit provides all the essential components with an optimized cell-labeling protocol. It is suitable for proliferating and non-proliferating cells, and can be used for both suspension and adherent cells. Using 100 uL of reagents per well in a 96-well format, this kit provides sufficient reagents to perform 200 assays. Using 25 uL of reagents per well in a 384-well format, this kit provides sufficient reagents to perform 800 assays.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 22787 | 200 Tests | Price |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 635 nm |
| Emission | 670 nm |
| Cutoff | 665 nm |
| Recommended plate | Black wall/clear bottom |
| Instrument specification(s) | Bottom read mode |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 10, 2026
