Screen Quest™ No Wash Potassium Channel Assay Kit
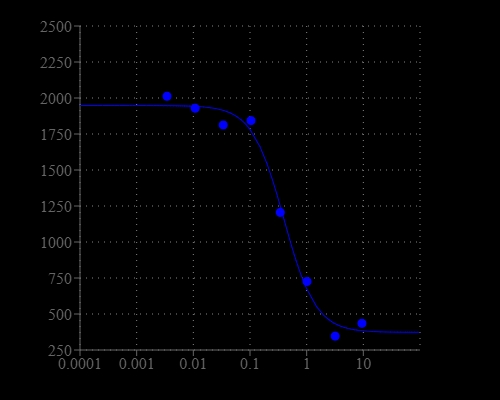
Potassium (K+) ion channel plays an important role in regulating fundamental biological processes including heart rate, hormone and neurotransmitter secretion, water and electrolyte balance. Potassium ion channel has been considered as drug targets for disease indications including arrhythmia, pain, diabetes, neurological dysfunctions etc. The permeability of Tl+ through K+ channel has been widely used to assay K+ channel. The cells that express K+ channel of interests (e.g. hERG, Kv1.3, Kir2.1, KATP) are pre-loaded with a Tl+ sensitive dye. The dye is non-fluorescent and is permeable to cell membrane. Once inside the cell, the non-fluorescent AM ester dye is cleaved by endogenous esterase into a negatively charges dye that stays inside cells. When a stimulus buffer containing low dose of Tl+ is added to cells, the Tl+ flows across the K+ channel and binds to Tl+ sensitive dye, generating a fluorescent signal. This signal is proportional to the activity of K+ channel. If an antagonist or antagonist is added to the cells, the fluorescent signal decreases or increases respectively, to reflect the inhibited or stimulated activity of K+ channel.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 36550 | 1 Plate | Price | |
| 36551 | 10 Plates | Price | |
| 36552 | 100 Plates | Price |
Spectral properties
| Excitation (nm) | 494 |
| Emission (nm) | 516 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 490 nm |
| Emission | 525 nm |
| Cutoff | 515 nm |
| Recommended plate | Black wall/clear bottom |
| Instrument specification(s) | Bottom read mode/Programmable liquid handling |
| Other instruments | ArrayScan, FDSS, FLIPR, FlexStation, IN Cell Analyzer, NOVOStar, ViewLux |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on April 28, 2026

