Thiolite™ Blue, AM
Example protocol
PREPARATION OF STOCK SOLUTIONS
Unless otherwise noted, all unused stock solutions should be divided into single-use aliquots and stored at -20 °C after preparation. Avoid repeated freeze-thaw cycles
Prepare a 1 to 5 mM stock solution of Thiolite™ Blue in high-quality, anhydrous DMSO.
PREPARATION OF WORKING SOLUTION
On the day of the experiment, either dissolve Thiolite™ Blue AM in DMSO or thaw an aliquot of the indicator stock solution to room temperature. Prepare a 1X Thiolite™ Blue AM working solution in a buffer of your choice (e.g., Hanks and Hepes buffer, pH 7), and mix them well by vortexing.
Note: The final concentration of the dye working solution should be empirically determined for different cell types and/or experimental conditions. It is recommended to test concentrations that are at least over a ten-fold range. The recommended concentration in Jurkat cells is 1-10 µM.
SAMPLE EXPERIMENTAL PROTOCOL
The following is a recommended protocol for loading Thiolite™ Blue AM into live mammalian cells. This protocol only provides a guideline, should be modified according to your specific needs.
- Prepare viable cells as desired.
- Treat cells with test compounds.
- Centrifuge the cells to get 1-5 × 105 cells per tube.
Resuspend cells in 1 mL of Thiolite™ Blue AM working solution.
Note: Alternatively, the DMSO stock solution can be added directly to the cells without removing the medium. For example, add 1 µL of 1 mM DMSO stock solution into 1 mL cells
- Incubate the dye-loaded plate in a cell incubator at 37 °C for 15 to 60 minutes.
- Remove the dye working solution and wash cells with HHBS or buffer of your choice to remove any excess probes.
- Resuspend the cells in 1 mL of pre-warmed HHBS or medium to get 2-10 × 105 cells per tube.
- Measure the fluorescence intensity using either a fluorescence microscope equipped with a DAPI filter set, a flow cytometer equipped with a UV/violet laser and a 450/40 nm filter (Pacific Blue channel), or a fluorescence plate reader at Ex/Em = 340/460 nm cutoff 420 nm.
Calculators
Common stock solution preparation
| 0.1 mg | 0.5 mg | 1 mg | 5 mg | 10 mg | |
| 1 mM | 301.859 µL | 1.509 mL | 3.019 mL | 15.093 mL | 30.186 mL |
| 5 mM | 60.372 µL | 301.859 µL | 603.719 µL | 3.019 mL | 6.037 mL |
| 10 mM | 30.186 µL | 150.93 µL | 301.859 µL | 1.509 mL | 3.019 mL |
Molarity calculator
| Mass (Calculate) | Molecular weight | Volume (Calculate) | Concentration (Calculate) | Moles | ||||
| / | = | x | = |
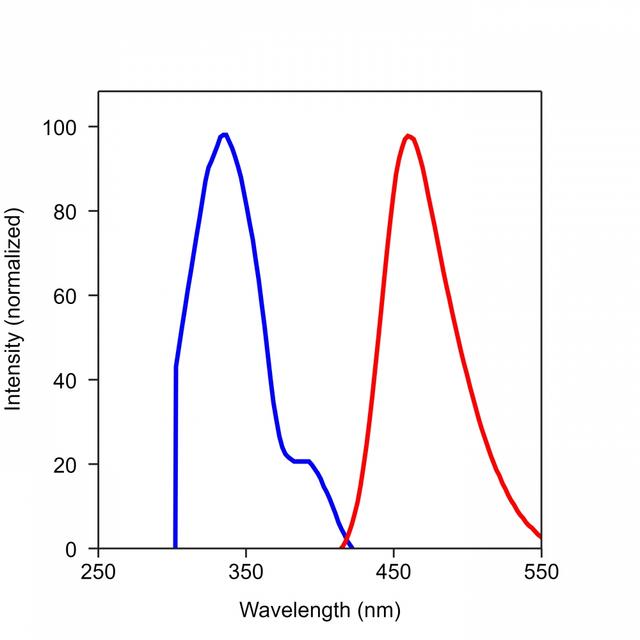
Spectrum
Citations
Authors: Xiao, Man and Du, Guankui and Zhong, Guobing and Yan, Dongjing and Zeng, Huazong and Cai, Wangwei
Journal: PloS one (2016): e0151103
Authors: Niikura, Kenichi and Sugimura, Naotoshi and Musashi, Yusuke and Mikuni, Shintaro and Matsuo, Yasutaka and Kobayashi, Shintaro and Nagakawa, Keita and Takahara, Shuko and Takeuchi, Chie and Sawa, Hirofumi and others, undefined
Journal: Molecular biosystems (2013): 501--507
References
Authors: Meziane D, Barras A, Kromka A, Houdkova J, Boukherroub R, Szunerits S.
Journal: Anal Chem (2012): 194
Authors: Li Y, Duan Y, Li J, Zheng J, Yu H, Yang R.
Journal: Anal Chem. (2012)
Authors: Peer CJ, Spencer SD, VanDenBerg DA, Pacanowski MA, Horenstein RB, Figg WD.
Journal: J Chromatogr B Analyt Technol Biomed Life Sci (2012): 132
Authors: Onuegbu J, Fu A, Glembocki O, Pokes S, Alexson D, Hosten CM.
Journal: Spectrochim Acta A Mol Biomol Spectrosc (2011): 456
Authors: Guo XF, Zhao PX, Wang H, Zhang HS.
Journal: J Chromatogr B Analyt Technol Biomed Life Sci (2011): 3932