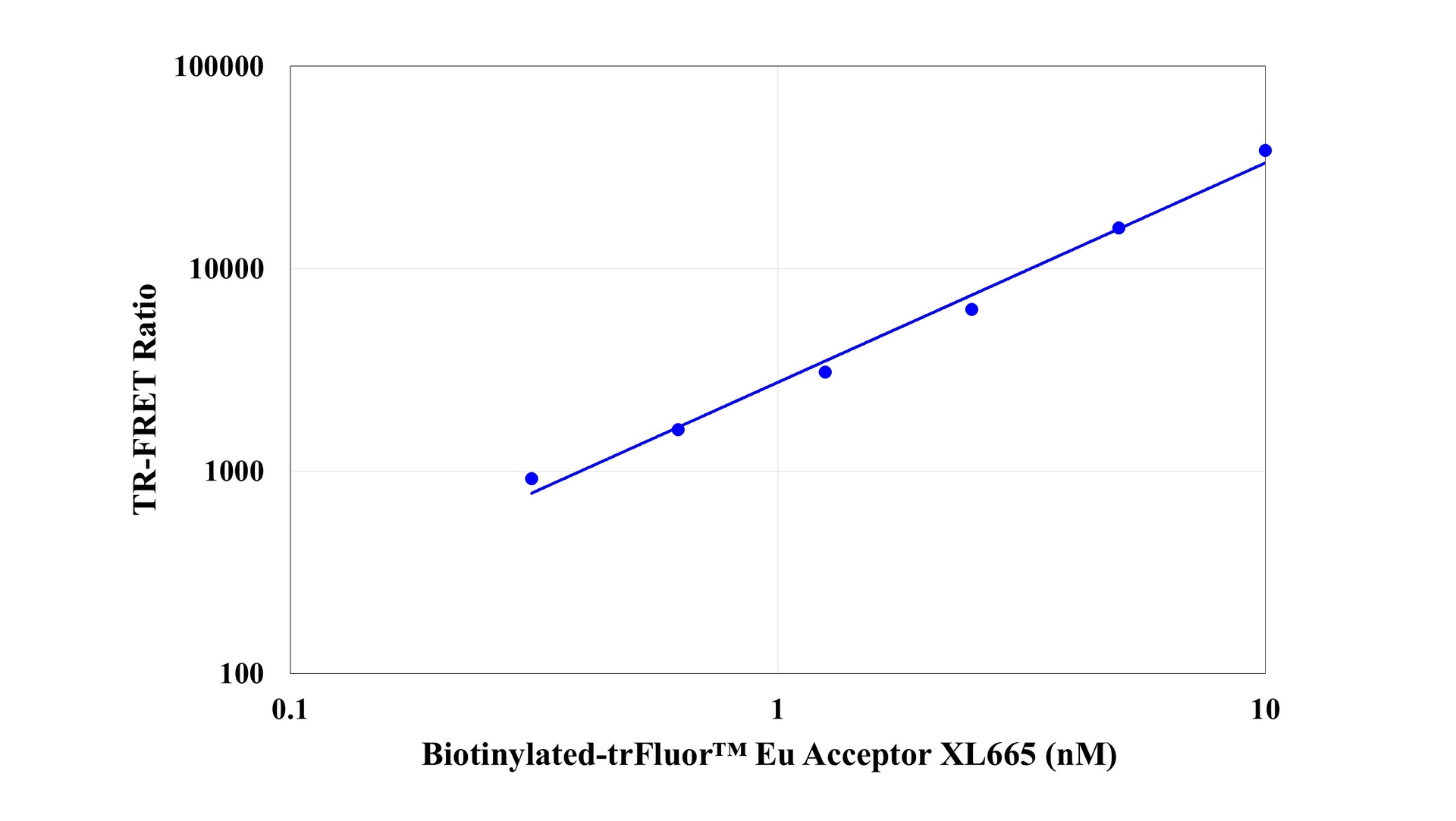
trFluor™ Eu-streptavidin conjugate
Streptavidin conjugates are widely used together with a conjugate of biotin for specific detection of a variety of proteins, protein motifs, nucleic acids and other molecules since streptavidin has a very high binding affinity for biotin. This trFluor™ Eu-streptavidin conjugate comprises streptavidin (as the biotin-binding protein) with trFluor™ Eu covalently attached (as the time-resolved red fluorescent europium label). It is commonly used as a second step reagent for indirect immunofluorescent staining, when used in conjunction with biotinylated primary antibodies. It is a very valuable tool for biotin-streptavidin-based biological assays and tests using TR-FRET platform. A variety of the complementary biotinylated reagents are available from numerous commercial vendors.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 16925 | 100 ug | Price |
Physical properties
| Molecular weight | ~52000 |
| Solvent | Water |
Spectral properties
| Correction factor (260 nm) | 0.911 |
| Correction factor (280 nm) | 0.777 |
| Extinction coefficient (cm -1 M -1) | 21000 |
| Excitation (nm) | 298 |
| Emission (nm) | 617 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 25, 2026

