Amplite® Fluorimetric Glutamic Acid Assay Kit *Red Fluorescence*
| Price | |
| Catalog Number | |
| Unit Size | |
| Quantity |
| Telephone | 1-800-990-8053 |
| Fax | 1-800-609-2943 |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Shipping | Standard overnight for United States, inquire for international |
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12171501 |
| Overview |
Platform
Fluorescence microplate reader
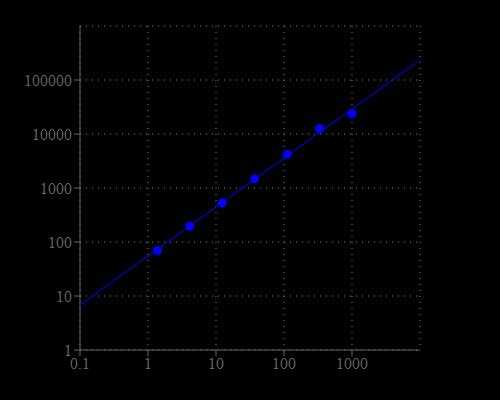
| Excitation | 540 nm |
| Emission | 590 nm |
| Cutoff | 570 nm |
| Recommended plate | Solid black |
Components
Example protocol
AT A GLANCE
Protocol summary
- Prepare Glutamic Acid working solution (50 µL)
- Add Glutamic Acid standards and/or test samples (50 µL)
- Incubate at room temperature for 30 minutes - 2 hours
- Monitor fluorescence intensity at Ex/Em = 540/590 nm (Cutoff = 570 nm)
Important notes
Thaw all the kit components at room temperature before starting the experiment.
PREPARATION OF STOCK SOLUTION
1. NADP stock solution (200X):
Add 100 µL of Dilution Buffer (Component E) into the vial of NADP (Component C) to make 200X NADP stock solution.
2. Glutamic Acid standard solution (100 mM):
Add 200 µL of Dilution Buffer (Component E) into the vial of Glutamic Acid (Component D) to make 100mM Glutamic Acid standard solution.
PREPARATION OF STANDARD SOLUTION
For convenience, use the Serial Dilution Planner: https://www.aatbio.com/tools/serial-dilution/10054
Take 100 mM Glutamic Acid standard solution and perform 1:100 in Dilution Buffer (Component E) to make 1000 µM Glutamic Acid standard solution (SD7). Take 1000 µM Glutamic Acid standard solution (SD7) and perform 1:3 serial dilutions to get serially diluted Glutamic Acid standards (SD6 - SD1) with Dilution Buffer (Component E).
PREPARATION OF WORKING SOLUTION
1. Add 10 mL of Assay Buffer (Component B) into the bottle of Enzyme Mix (Component A).
2. Add 50 µL 200X NADP stock solution into the Enzyme Mix bottle, and mix well to make Glutamic Acid working solution. Note: This Glutamic Acid working solution is enough for two 96-well plates. It is unstable at room temperature, and should be used promptly within 2 hours and avoid exposure to light. Note: Alternatively, one can make a 50X of Enzyme Mix stock solution by adding 200 μL of H2O into the bottle of Enzyme Mix (Component A), and then prepare the Glutamic Acid working solution by mixing the stock solution with Assay Buffer (Component B) and 200X NADP stock solution proportionally.
SAMPLE EXPERIMENTAL PROTOCOL
Table 1. Layout of Glutamic Acid standards and test samples in a solid black 96-well microplate. SD = Glutamic Acid Standard, BL = Blank Control, TS = Test Sample.
| BL | BL | TS | TS |
| SD1 | SD1 | ... | ... |
| SD2 | SD2 | ... | ... |
| SD3 | SD3 | ||
| SD4 | SD4 | ||
| SD5 | SD5 | ||
| SD6 | SD6 | ||
| SD7 | SD7 |
Table 2. Reagent composition for each well
| Well | Volume | Reagent |
| SD1-SD7 | 50 µL | Serial Dilution (1 to 1000 µM) |
| BL | 50 µL | Dilution Buffer (Component E) |
| TS | 50 µL | Sample |
- Prepare Glutamic Acid standards (SD), blank controls (BL), and test samples (TS) according to the layout provided in Tables 1 and 2. For a 384-well plate, use 25 µL of reagent per well instead of 50 µL.
- Add 50 µL of Glutamic Acid working solution into each well of Glutamic Acid standard, blank control, and test samples to make the total Glutamic Acid assay volume of 100 µL/well. For a 384-well plate, add 25 µL of Glutamic Acid working solution into each well intead, for the total volume of 50 µL/well.
- Incubate the reaction at room temperature for 30 minutes to 2 hours, protected from light.
- Monitor the fluorescence increase with a fluorescence plate reader at Excitation = 530 - 570 nm, Emission = 590 - 600 nm (optimal Ex/Em = 540/590 nm), Cutoff = 570 nm. Note: The contents of the plate can also be transferred to a white clear bottom plate and read by an absorbance microplate reader at the absorbance ratio of ~570 nm to ~605 nm (A575nm/A605nm). The absorption detection has lower sensitivity compared to the fluorescence reading.
Images
Citations
Authors: Alexander, Ashley M and Luu, Justin M and Raghuram, Vishnu and Bottacin, Giulia and van Vliet, Simon and Read, Timothy D and Goldberg, Joanna B
Journal: Microbiology (2024): 001445
Authors: Xuan, Zhao and Barthet, Gael and Shioi, Junichi and Xu, Jindong and Georgakopoulos, Anastasios and Bruban, Julien and Robakis, Nikolaos K
Journal: Journal of Biological Chemistry (2013): 30495--30501
References
Authors: Hillman M, Torn C, L and in-Olsson M., undefined
Journal: Clin Exp Immunol (2009): 255
Authors: Daka B, Svensson MK, Lernmark K, Mincheva-Nilsson L, Hallmans G, Rol and sson O., undefined
Journal: Autoimmunity (2009): 507
Authors: AuCoin DP, Sutherl and MD, Percival AL, Lyons CR, Lovchik JA, Kozel TR.
Journal: Diagn Microbiol Infect Dis (2009): 229
Authors: Blanc F, Ruppert E, Kleitz C, Valenti MP, Cretin B, Humbel RL, Honnorat J, Namer IJ, Hirsch E, Manning L, de Seze J.
Journal: J Neurol Sci (2009): 69
Authors: Liu H, Li S, Zhang Y, Yan Y, Li Y.
Journal: Acta Biochim Biophys Sin (Shanghai) (2009): 545
Authors: Errichiello L, Perruolo G, Pascarella A, Formisano P, Minetti C, Striano S, Zara F, Striano P.
Journal: J Neuroimmunol (2009): 120
Authors: Gos T, Gunther K, Bielau H, Dobrowolny H, Mawrin C, Trubner K, Brisch R, Steiner J, Bernstein HG, Jankowski Z, Bogerts B.
Journal: J Affect Disord (2009): 45
Authors: Chen D, He BB, Zhao DJ, Jiang QY, Wang ZR, Zhou J, Yu H, Wang QQ, Tang GP.
Journal: Zhejiang Da Xue Xue Bao Yi Xue Ban (2009): 31
Authors: Lee DY, Chun JH, Ha HJ, Park J, Kim BS, Oh HB, Rhie GE.
Journal: FEMS Immunol Med Microbiol (2009): 165
Authors: Virgilio R, Corti S, Agazzi P, Santoro D, Lanfranconi S, C and elise L, Bresolin N, Comi GP, Bersano A.
Journal: J Neurol Neurosurg Psychiatry (2009): 95
Application notes
Design of potent inhibitors of acetylcholinesterase using morin as the starting compound
Acetylcholinesterase Inhibitory Activity of Pigment Echinochrome A
Induction of Neurite Outgrowth in PC12 Cells
Induction of Neuritogenesis in PC12 Cells by a Pulsed Electromagnetic Field
FAQ
What are the similarities between liposomal glutathione and reduced glutathione?
Why should I use an absorbance ratio at A575nm/A605nm when using most of your Amplite® Colorimetric Assay Kits?
How should I reconstitute an NADPH standard?
Will Amplite® Fluorimetric NAD/NADH Ratio Assay Kit *Red Fluorescence* work with NADP/NADPH? Can this kit measure NADP+ and NADPH?