Amplite® Fluorimetric D-Lactate Dehydrogenase (LDH) Assay Kit
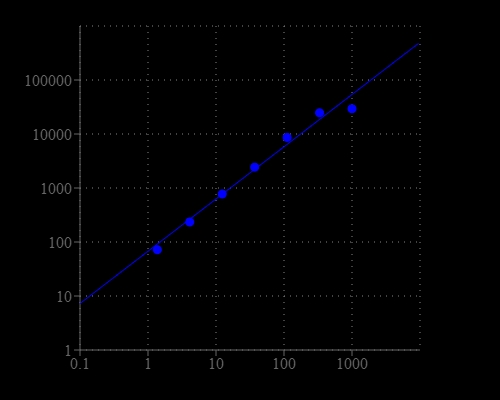
Lactate dehydrogenase (LDH) is an oxidoreductase enzyme that catalyzes the interconversion of pyruvate and lactate. LDH is present in cytosol of a wide variety of organisms, including animals and plants. Cells release LDH into the bloodstream after tissue damage or red blood cell hemolysis. Since LDH is a fairly stable enzyme, it has been widely used to evaluate the presence of damage and toxicity of tissue and cells. Quantification of LDH has a broad range of applications. LDH is also elevated in certain pathological conditions such as cancer. This Amplite® Lactate Dehydrogenase Assay Kit provides a fluorescence-based method for detecting D-lactate dehydrogenase (D-LDH) in biological samples such as serum, plasma, urine, as well as in cell culture samples. In the enzyme coupled assay, LDH is proportionally related to the concentration of NADH that is specifically monitored by a fluorogenic NADH sensor. This assay is specific for D-LDH. The fluorescence signal can be read by a fluorescence microplate reader. With this fluorimetric Amplite® D-lactate Dehydrogenase Assay Kit, we were able to detect as little as 1 mU/mL D-lactate dehydrogenase in a 100 µL reaction volume.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 13808 | 200 Tests | Price |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 540 nm |
| Emission | 590 nm |
| Cutoff | 570 nm |
| Recommended plate | Solid black |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 4, 2026
