Amplite® Fluorimetric Goat Anti-Mouse IgG-HRP Conjugate ELISA Assay Kit
Red Fluorescence
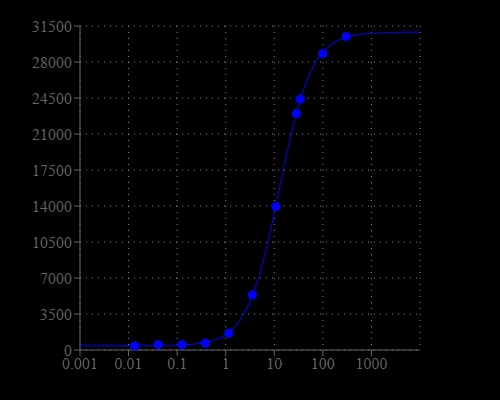
Horseradish Peroxidase (HRP) is a small molecule (MW ~40 KD) that is widely used in a variety of biological detections. HRP conjugates are extensively used as secondary detection reagents in ELISAs, immuno-histochemical techniques, Northern, Southern and Western blot analyses. Due to its small size, it rarely causes steric hindrance problem with antibody/antigen complex formation. It is usually conjugated to an antibody in a 4:1 ratio. Additionally, HRP is inexpensive compared to other labeling enzymes. The major disadvantage associated with peroxidase is their low tolerance to many preservatives such as sodium azide that inactivates peroxidase activity even at low concentration. Our Amplite® Fluorimetric ELISA Assay Kit contains all the essential components including our fluorogenic Amplite® Red HRP substrate for ELISA detection. The kit provides an optimized assay protocol that is compatible with HTS liquid handling, as little as 10 pg of a monoclonal antibody/well of a microplate can be detected. Its signal can be easily read by either fluorescence microplate reader or absorbance microplate reader. It can be used for assays that detect goat anti-mouse IgG as the secondary detection agent.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 11540 | 1000 Tests | Price |
Spectral properties
| Excitation (nm) | 571 |
| Emission (nm) | 584 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12171501 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 540 nm |
| Emission | 590 nm |
| Cutoff | 570 nm |
| Recommended plate | Solid black |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 5, 2026

