Bucculite™ Flow Cytometric XdU Cell Proliferation Assay Kit
Red Laser-Comptatible
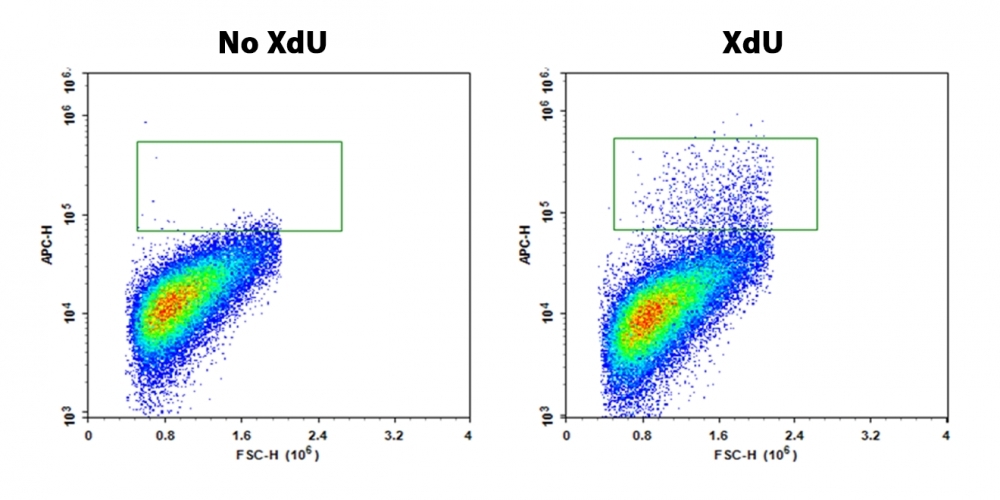
Monitoring cell proliferation is one of the most reliable methods to assess cell viability, cell cycles and genotoxicity. An essential way to detect cell proliferation is to measure DNA synthesis in the presence of thymidine during the S-phase of cells growth. Bucculite™ Flow Cytometric XdU Cell Proliferation Assay Kit uses XdU which is incorporated into cellular DNA during DNA synthesis. After fixing cells, the incorporated XdU is labelled with iFluor® 647 MTA. The resulted iFluor® 647-labeled DNA formed in cells is visualized in Cy5 Channel. Bucculite™ Flow Cytometric XdU Cell Proliferation Assay Kit provides an alternative to anti-BrdU antibody-based assay and EdU click chemistry assay. It is sensitive and might be used for measuring active DNA synthesis at single-cell level.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 22325 | 100 Tests | Price |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12171501 |
Instrument settings
| Flow cytometer | |
| Excitation | 633/640 nm laser |
| Emission | 660/20 nm filter |
| Instrument specification(s) | APC channel |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 17, 2026
