Cell Meter™ Annexin V Binding Apoptosis Assay Kit
Green Fluorescence Excited at 405 nm
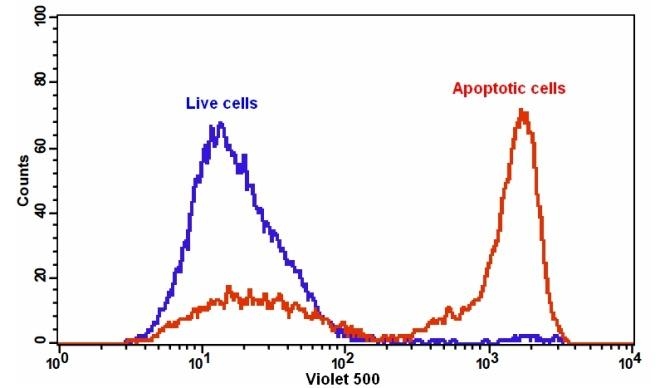
Our Cell Meter™ assay kits are a set of tools for monitoring cell viability. There are a variety of parameters that can be used for monitoring cell viability. This particular kit is designed to monitor cell apoptosis through measuring the translocation of phosphatidylserine (PS). In apoptosis, PS is transferred to the outer leaflet of the plasma membrane. The appearance of phosphatidylserine on the cell surface is a universal indicator of the initial/intermediate stages of cell apoptosis and can be detected before morphological changes can be observed. This kit uses a fluorescent Annexin V that specifically binds PS. Annexin V conjugates have been demonstrated to selectively bind PS. This particular assay kit is optimized to monitor cell apoptosis using a flow cytometer with the 405 laser with 525/40 emission filter set.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 22829 | 100 Tests | Price |
Spectral properties
| Absorbance (nm) | 412 |
| Correction factor (260 nm) | 0.769 |
| Correction factor (280 nm) | 0.365 |
| Extinction coefficient (cm -1 M -1) | 25000 1 |
| Excitation (nm) | 410 |
| Emission (nm) | 501 |
| Quantum yield | 0.81 1 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Instrument settings
| Flow cytometer | |
| Excitation | 405 nm laser |
| Emission | 525/40 nm filter |
| Instrument specification(s) | AmCyan channel |
| Fluorescence microscope | |
| Excitation | Violet filter set |
| Emission | Violet filter set |
| Recommended plate | Black wall/clear bottom |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on October 8, 2024

