Cell Meter™ Caspase 8 Activity Apoptosis Assay Kit
Red Fluorescence
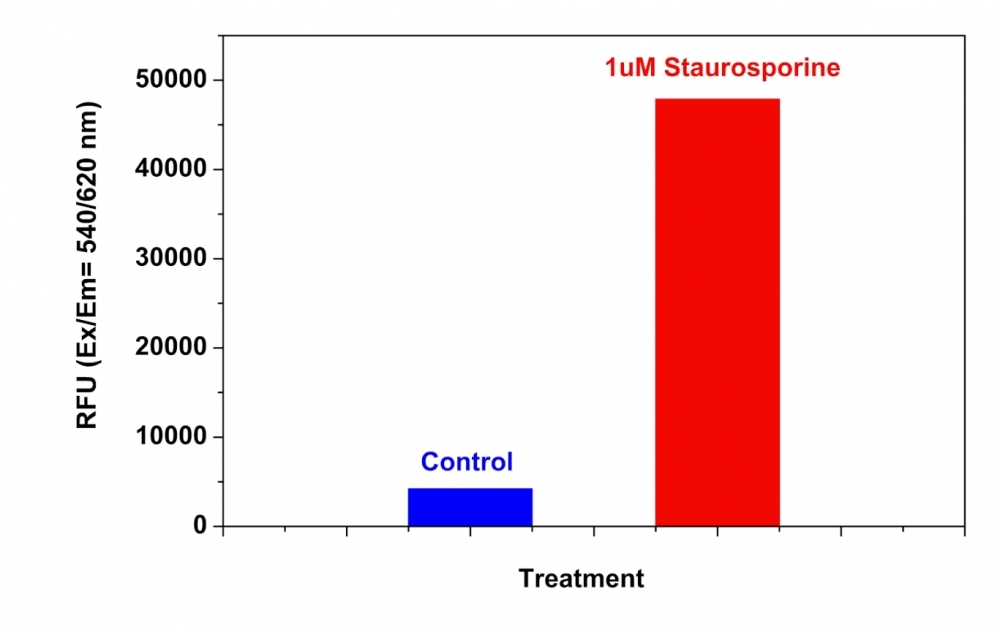
Our Cell Meter™ assay kits are a set of tools for monitoring cellular functions. There are a variety of parameters that can be used to monitor cell apoptosis. This particular kit is designed to monitor cell apoptosis by measuring caspase 8 activity. Caspase 8 is a caspase protein, encoded by the CASP8 gene. Caspase 8 also plays an important role in neurodegenerative diseases such as Huntington disease. Caspase 8 is proven to have substrate selectivity for the peptide sequence Ile-Glu-Thr-Asp (IETD). This kit uses (Ac-IETD)-ProRed™ as a fluorogenic indicator for caspase 8 activity. Cleavage of ProRed™ peptides by caspase 8 generates strongly fluorescent ProRed™. The kit provides all the essential components with an optimized assay protocol. The assay can be readily adapted for high throughput screenings. It can be used to either quantify the activated caspase 8 activities in apoptotic cells or screen the caspase 8 inhibitors.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 22816 | 100 Tests | Price |
Spectral properties
| Excitation (nm) | 532 |
| Emission (nm) | 619 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 540 nm |
| Emission | 620 nm |
| Cutoff | 610 nm |
| Recommended plate | Black wall/clear bottom |
| Instrument specification(s) | Top or bottom read mode |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 5, 2026

