Cell Meter™ No Wash and Probenecid-Free Endpoint Calcium Assay Kit
Optimized for microplate reader
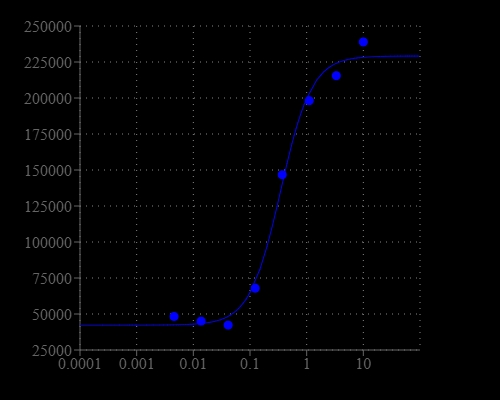
Cell Meter™ No Wash and Probenecid-Free Endpoint Calcium Assay Kit enables homogeneous fluorescence-based assays for detecting intracellular calcium mobilization without the need to use kinetics reading mode. It can be used on Fluorescence microplate readers with bottom read mode that do not have a build-in liquid dispenser or the ability of kinetics reading. After loading the Fluo-8E™AM dye into cells of interest, without wash steps, one can simply add the calcium flux agonist by a liquid dispenser or hand pipetting, and then read the plate by a fluorescent reader. Fluo-8E™ AM can cross cell membrane passively by diffusion. Once inside the cells, the lipophilic blocking groups of Fluo-8E™ AM are cleaved by esterase, resulting in a negatively charged fluorescent dye that stays inside cells. Its fluorescence is greatly enhanced and long lasting upon binding to calcium. When cells expressing GPCR of interest are stimulated with an agonist, the receptor signals the release of intracellular calcium, which significantly increases the fluorescence of Fluo-8E™. The characteristics of its high sensitivity, >100 times fluorescence enhancement and long lasting fluorescent signal make Fluo-8E™ an ideal indicator for the measurement of cellular calcium on fluorescence microplate readers that do not have the fluid transfer and kinetic reading mode capability. The Cell Meter™ No Wash and Probenecid-Free Endpoint Calcium Assay Kit can be performed in a 96-well or 384-well microtiter-plate format.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 36312 | 100 Tests | Price |
Spectral properties
| Correction factor (260 nm) | 1.076 |
| Correction factor (280 nm) | 0.769 |
| Extinction coefficient (cm -1 M -1) | 23430 |
| Excitation (nm) | 495 |
| Emission (nm) | 516 |
| Quantum yield | 0.16 1 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 490 nm |
| Emission | 525 nm |
| Cutoff | 515 nm |
| Recommended plate | Black wall/Clear bottom |
| Instrument specification(s) | Bottom read mode |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on April 30, 2026

