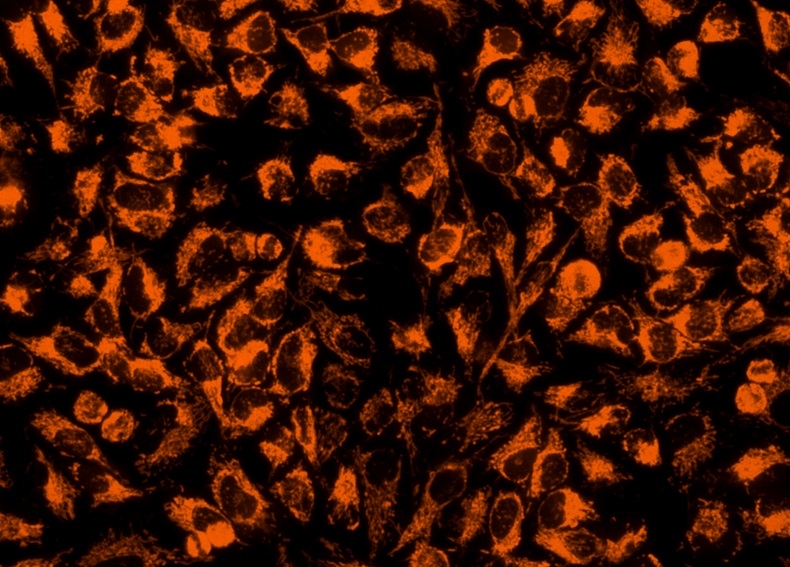
Cell Navigator® Mitochondrion Staining Kit
Orange Fluorescence with 405 nm Excitation
Our Cell Navigator® fluorescence imaging kits are a set of fluorescence imaging tools for labeling sub-cellular organelles such as membranes, lysosomes, mitochondria, nuclei, etc. The selective labeling of live cell compartments provides a powerful method for studying cellular events in a spatial and temporal context. This particular kit is designed to label mitochondria of live cells in orange fluorescence of large Stokes Shift. The kit uses a proprietary dye that selectively accumulates in mitochondria probably via the mitochondrial membrane potential gradient. The mitochondrial indicator, a hydrophobic compound, easily permeates intact live cells and trapped in mitochondria after it gets into cells. This fluorescent mitochondrial indicator is retained in mitochondria for a long time since it carries a cell-retaining group. This key feature significantly increases the staining efficiency. The mitochondrial stain used in the kit is quite photostable, making the fluorescence quite robust. The kit provides all the essential components with an optimized cell-labeling protocol. It is suitable for proliferating and non-proliferating cells, and can be used for both suspension and adherent cells.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 22673 | 500 Assays | Price |
Spectral properties
| Excitation (nm) | 425 |
| Emission (nm) | 522 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Instrument settings
| Fluorescence microscope | |
| Excitation | Violet filter |
| Emission | Violet filter |
| Recommended plate | Black wall/clear bottom |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 10, 2026

