CytoFix™ Red Mitochondrial Stain
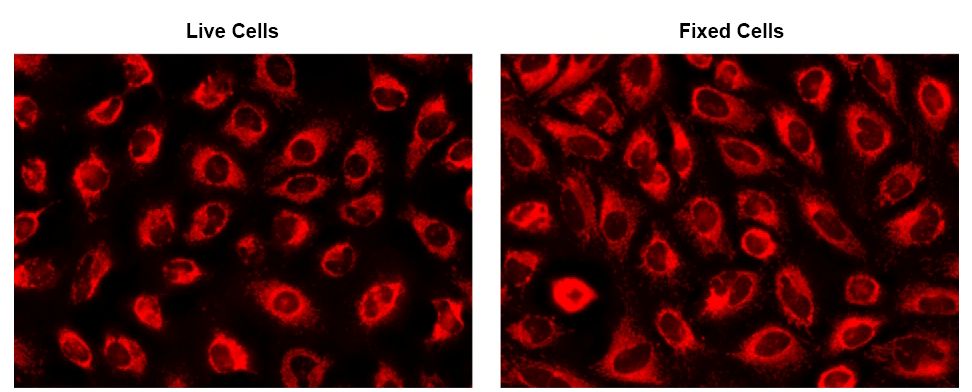
CytoFix™ Red mitochondrial stain is a dye that selectively stains mitochondria independent of mitochondrial membrane potential gradient. Due to this functionality, CytoFix™ Red mitochondrial stain is well retained in mitochondria even after fixation. The dye permeates intact live cells and gets trapped in live cells. Its key features include high staining efficiency, long retention after fixation with minimal hands on time. CytoFix™ Red mitochondrial stain can be used with GFP expressed cells without overlapping the fluorescence of GFP, making it useful for multiplexing analysis. It can be used for both suspension and adherent cells and readily adapted for a wide variety of fluorescence platforms.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 23200 | 500 Tests | Price |
Physical properties
| Molecular weight | N/A |
| Solvent | DMSO |
Spectral properties
| Excitation (nm) | 564 |
| Emission (nm) | 651 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12171501 |
| CAS | N/A |
Instrument settings
| Fluorescence microscope | |
| Excitation | Cy3/TRITC filter set |
| Emission | Cy3/TRITC filter set |
| Recommended plate | Black wall/clear bottom |
| Instrument specification(s) | Cy3/TRITC filter set |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 1, 2026

