FluoroQuest™ PLUS Antifade Mounting Medium
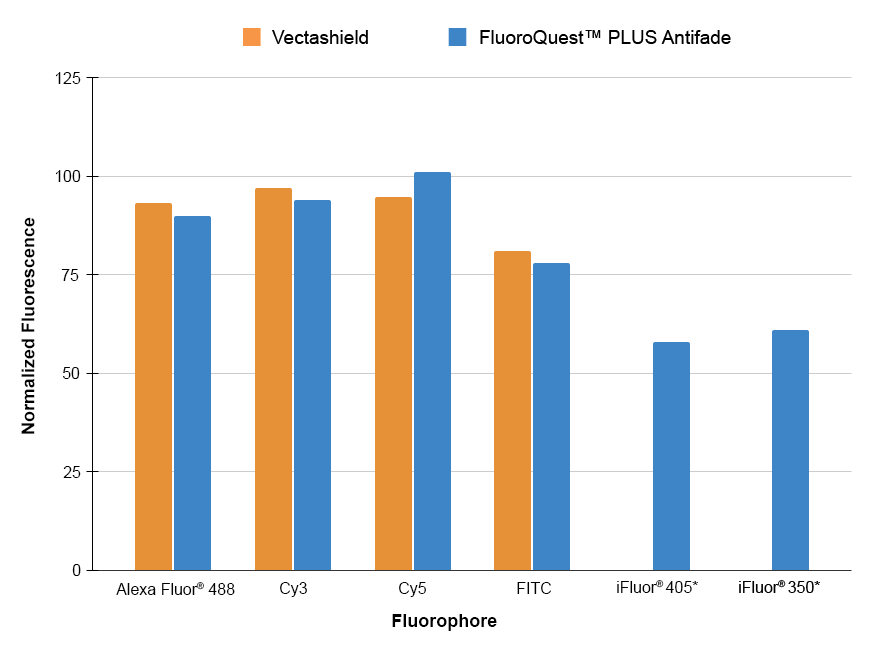
FluoroQuest™ PLUS Antifade Mounting Medium, the latest breakthrough in non-setting media technology, provides unparalleled protection against fading throughout the visible spectrum, including far-red wavelengths. Its compatibility with most commercially available fluorophores makes it a versatile solution for various fluorescence imaging applications. With its improved formulation, it outperforms its predecessor, the FluoroQuest™ Antifade Mounting Medium, by exhibiting no inherent background or toning while ensuring superior retention of fluorophore signals across the spectrum, including far-red wavelengths. Additionally, this superior mounting medium enables mounted sections to be viewed in just one hour, while its non-setting formulation ensures consistent and artifact-free results.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 20008 | 5 mL | Price |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 3, 2026
