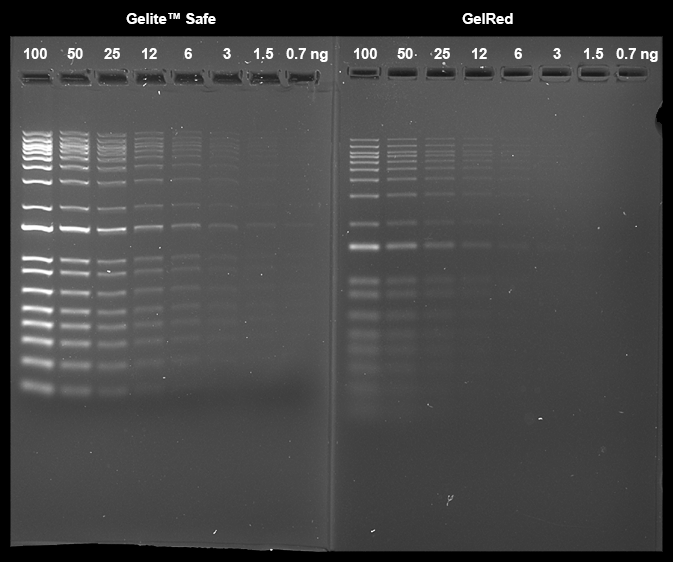
AAT Bioquest is committed to designing our products to be environment-friendly. It is part of how we enable our customers to make the world healthier, cleaner, and safer. Ethidium bromide (EtBr) has been commonly used as a DNA stain for many years. However, EtBr is harmful if swallowed and is very toxic if inhaled. EtBr has been shown to be mutagenic in various tests and is an aquatic toxin. SYBR® Safe was introduced as a safer alternative to EtBr and SYBR® Green, but unfortunately, it is much less sensitive than SYBR® Green. It only has sensitivity comparable to EtBr. Gelite™ Safe has been developed specifically to be less hazardous than EtBr for staining DNA in agarose and acrylamide gels with much higher sensitivity. Gelite™ Safe has greatly improved safety and uncompromised sensitivity. The exceptional sensitivity and strong DNA binding affinity of Gelite™ Safe allows DNA to be stained prior to or post electrophoresis without destaining. In addition to its superior binding properties, Gelite™ Safe is essentially non-fluorescent in the absence of nucleic acids showing very low background fluorescence. Upon binding to nucleic acids, Gelite™ Safe exhibits a considerable fluorescence enhancement by several orders of magnitude greater than that of EtBr. Gelite™ Safe was optimized to be compatible with various instruments, including UV and blue-light transiluminators, gel documentation systems, and laser scanners. It is the first single formulation that can be used in either the green or red channel at your preference. Unlike the membrane-permeant SYBR® Green, which is highly toxic to cells and the environment, the membrane-impermeant properties of Gelite™ Safe make it a much safer and noncytotoxic alternative. Furthermore, Ames testing has confirmed Gelite™ Safe to be significantly less mutagenic than EtBr and SYBR® Green, even at concentrations well above the working concentration used for gel staining. Ames mutagenicity test was performed in a dose-dependent manner for all test dyes pretreated with an S9 fraction from rat liver (SYBR® is a trademark of ThermoFisher).

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 17709 | 500 uL | Price |
| Molecular weight | N/A |
| Solvent | Water |
| Absorbance (nm) | 509 |
| Excitation (nm) | 513 |
| Emission (nm) | 552 |
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| Gel Imager | |
| Excitation | UV Transilluminator/Blue laser |
| Emission | SYBR® filter, GelStar® filter, GelGreen® filter, or GelRed® filter |
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |

