iFluor® Ultra 594 maleimide
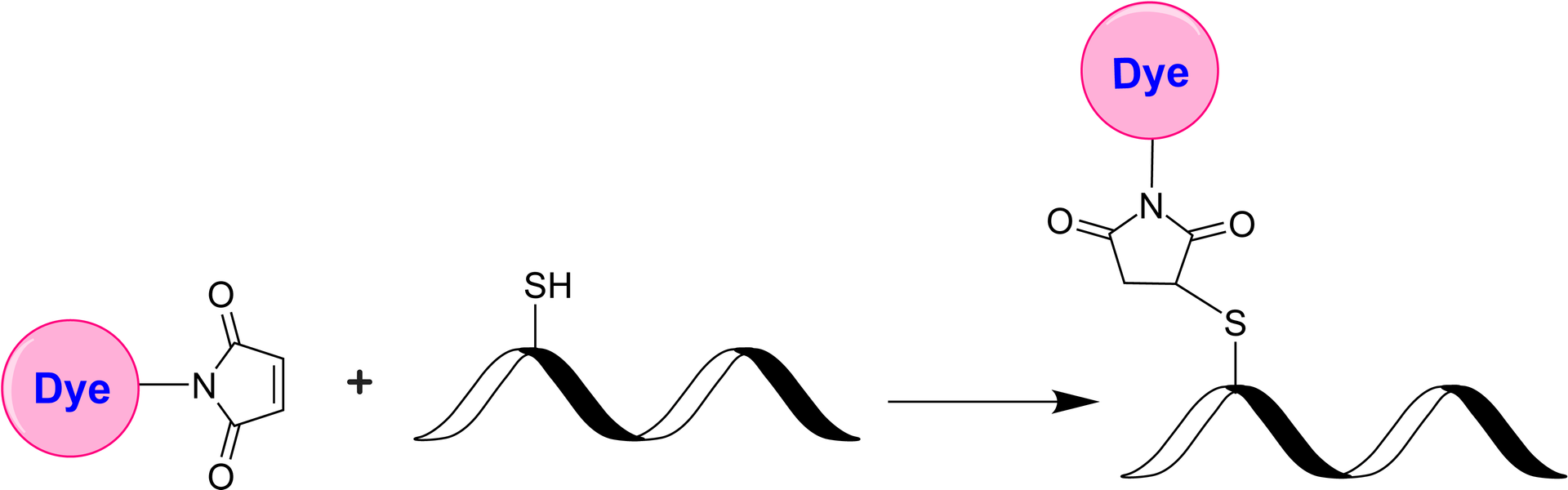
The iFluor® Ultra series represents an enhancement of our established iFluor® dyes, optimized for antibody labeling in fluorescence imaging and flow cytometry. Within this series, iFluor® Ultra 594 is an exceptionally bright red fluorescent dye excitable by 561 nm or 594 nm laser lines, with a peak emission at 601 nm. Antibody conjugates with iFluor® Ultra 594 exhibit superior brightness compared to those labeled with Alexa Fluor® 594 under identical conditions. Additionally, iFluor® Ultra 594 maintains stable fluorescence across a pH range of 4 to 10. The maleimide derivative of iFluor® Ultra 594 is widely used for conjugation to thiol groups on proteins, oligonucleotide thiophosphates, or low molecular weight ligands. Conjugates with iFluor® Ultra 594 demonstrate higher fluorescence intensity and greater photostability compared to those with other spectrally similar fluorophores, enhancing their utility for advanced fluorescence applications. Fluorescent dye-conjugated antibodies are crucial for protein identification in various applications, including fluorescent cell imaging, flow cytometry, western blotting, and immunohistochemistry. The advantages of these conjugates include increased sensitivity, multiplexing capabilities, and ease of use, facilitating complex biological studies.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 71653 | 1 mg | Price |
Physical properties
| Molecular weight | 1097.12 |
| Solvent | DMSO |
Spectral properties
| Absorbance (nm) | 585 |
| Correction factor (260 nm) | 0.07 |
| Correction factor (280 nm) | 0.05 |
| Extinction coefficient (cm -1 M -1) | 180000 1 |
| Excitation (nm) | 586 |
| Emission (nm) | 600 |
| Quantum yield | 0.52 1 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12171501 |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 6, 2026

