MitoROS™ 580
Optimized for Detecting Reactive Oxygen Species (ROS) in Mitochondria
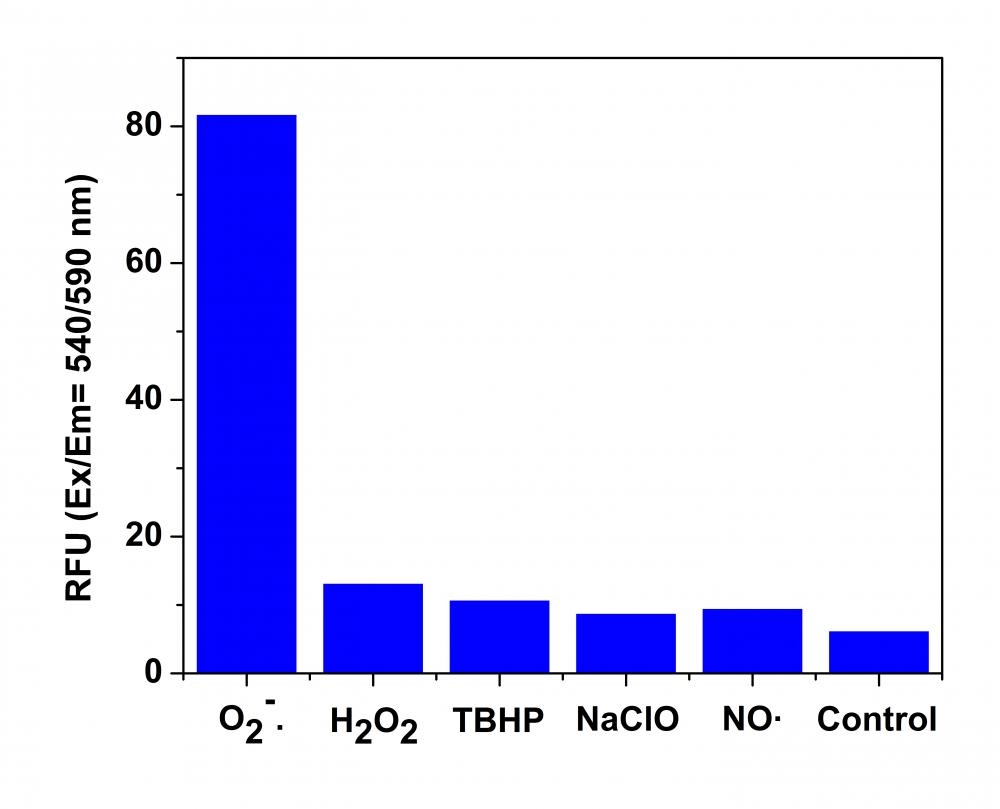
Reactive oxygen species (ROS) are chemically reactive molecules containing oxygen. Examples include superoxide, hydroxyl radical, singlet oxygen, and peroxides. ROS is highly reactive due to the presence of unpaired valence shell electrons. ROS forms as a natural byproduct of the normal metabolism of oxygen and has important roles in cell signaling and homeostasis. However, during times of environmental stress (e.g., UV or heat exposure), ROS levels can increase dramatically. This may result in significant damage to cell structures. Cumulatively, this is known as oxidative stress. MitoROS™ 580 is a superoxide-sensitive dye that is localized in mitochondria upon loading into live cells. Oxidation of MitoROS™ 580 by superoxide generates red fluorescence. MitoROS™ 580 can be used to monitor superoxide in mitochondria either with a fluorescence microscope or flow cytometer. MitoROS™ 580 reagent permeates live cells where it selectively targets mitochondria. It is rapidly oxidized by superoxide. It is less likely to be oxidized by other reactive oxygen species (ROS) and reactive nitrogen species (RNS). The oxidized product is highly fluorescent in cells. MitoROS™ 580 provides a valuable tool for investigating oxidative stress in various pathologies. MitoROS™ 580 is equivalent to the MitoSOX™ Mitochondrial Superoxide Indicator (#M36008) that is often used for live-cell imaging (MitoSOX™ is the trademark of ThermoFisher).

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 16052 | 500 Tests | Price |
Physical properties
| Molecular weight | N/A |
| Solvent | DMSO |
Spectral properties
| Excitation (nm) | 500 |
| Emission (nm) | 582 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 2, 2026

