Portelite™ Fluorimetric ssDNA Quantitation Kit
Optimized for Cytocite™ and Qubit™ Fluorometers
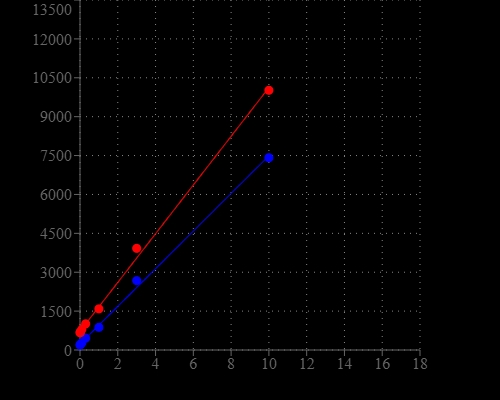
Portelite™ Fluorimetric ssDNA Quantitation Kit is designed to rapidly measure single-stranded DNA. The kit has all the essential reagents, including Helixyte™ Green ssDNA reagent, dilution buffer, and prediluted DNA standards. Helixyte™ Green ssDNA reagent is a sensitive fluorescent nucleic acid probe for quantifying oligonucleotides and single-stranded DNA (ssDNA) in solution. Simply dilute the reagent using the buffer provided, add your sample (any volume from 1–20 μL is acceptable), then read the concentration using CytoCite, Qubit® or other hand-held or desk top fluorometers (Qubit® is the trademark of ThermoFisher). The assay is accurate for initial sample concentrations from 50 pg/µL to 200 ng/µL, providing an assay range of 1–200 ng. The assay detects long oligonucleotides or ssDNA. Nucleotides and short oligo nucleotides of six bases or less do not interfere in the quantitation assay. The detection limit is not significantly interfered by the common contaminants in nucleic acid preparations, including salts, urea, ethanol, chloroform, detergents, proteins, nucleotides and short oligonucleotides of six bases. However, double-stranded DNA (dsDNA) and RNA do interfere with the assay as Helixyte™ Green ssDNA reagent binds to dsDNA and RNA to generate additional fluorescence signal. Portelite™ Fluorimetric ssDNA Quantitation Kit is optimized for CytoCite™ and Qubit® fluorometers.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 17625 | 200 Tests | Price |
Spectral properties
| Excitation (nm) | 498 |
| Emission (nm) | 519 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12171501 |
Instrument settings
| Qubit Fluorometer | |
| Excitation | 480 nm |
| Emission | 530 nm |
| Instrument specification(s) | 0.2 mL PCR vial |
| CytoCite Fluorometer | |
| Excitation | 480 nm |
| Emission | 530 nm |
| Instrument specification(s) | 0.2 mL PCR vial |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 6, 2026

