Thiolite™ Green
Thiolite™ Green is one of the most sensitive sensors for measuring thiol compounds. It gives a green fluorescent adduct upon reacting with thiol compounds (such as cysteine). It can be used to quantifying the number of cysteines on a protein. We have used it to measure glutathione fluorimetrically. It has >200-fold fluorescence enhancement upon reaction with thiol-containing compounds.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 21508 | 5 mg | Price |
Physical properties
| Molecular weight | ~400 |
| Solvent | DMSO |
Spectral properties
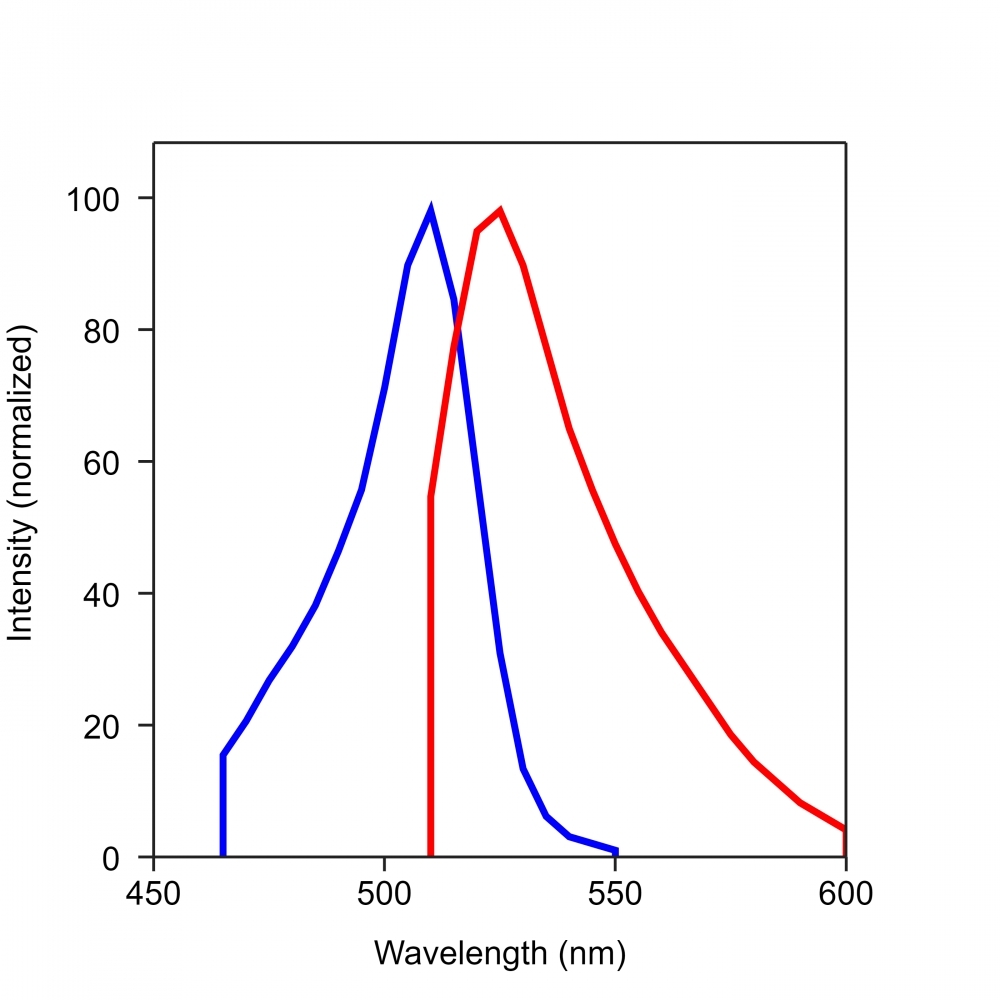
| Excitation (nm) | 505 |
| Emission (nm) | 524 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 27, 2026

