Tide Quencher™ 4WS alkyne
TQ4WS alkyne
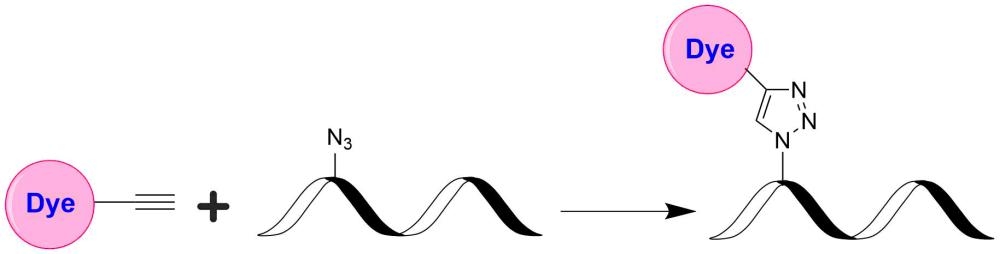
TQ4WS is designed to be a superior quencher to ROX, TF4, iFluor® 594, Alexa Fluor® 594 and Texas Red®. TQ4WS has (a). much stronger absorption; (b). much higher quenching efficiency; and (c). versatile reactive forms with desired solubility for labeling oligonucleotides and peptides. This TQ4WS-DBCO product is reactive to azides under copper-free conditions, and useful for click chemistry. DBCO is probably the most common alkyne among the strain promoted alkyne-azide cycloaddition (SPAAC), which is also termed as the Cu-free click reaction. Cyclooctynes and azides exclusively and efficiently react with each other while remain inert to naturally occurring functional groups such as amines. SPAAC enables labeling a wide variety of biomolecules without any auxiliary reagents in an aqueous and otherwise complex chemical environment through the formation of a stable triazole. DBCO (dibenzocyclooctynes) compounds comprise a class of reagents that possesses reasonably fast kinetics and good stability in aqueous buffers. Within physiological temperature and pH ranges, the DBCO group will not react with amines or hydroxyls that are naturally present in many biomolecules. Additionally, reaction of the DBCO group with the azide group is significantly faster than with sulfhydryl groups (–SH, thiol).

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 2069 | 1 mg | Price |
Physical properties
| Molecular weight | 1158.49 |
| Solvent | DMSO |
Spectral properties
| Absorbance (nm) | 605 |
| Correction factor (260 nm) | 0.149 |
| Correction factor (280 nm) | 0.136 |
| Extinction coefficient (cm -1 M -1) | 90000 1 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12171501 |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 5, 2026

