Tide Quencher™ 7WS azide
TQ7WS azide
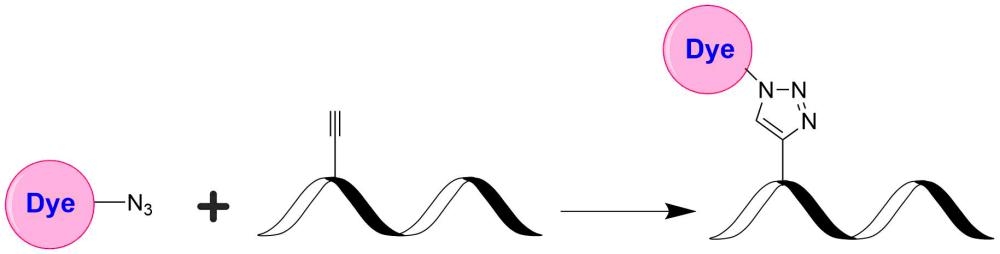
TQ7WS is designed to be a superior quencher to Cy7, TF7 and Alexa Fluor 750. TQ7WS has (a). much stronger absorption; (b). much higher quenching efficiency; and (c). versatile reactive forms with desired solubility for labeling oligonucleotides and peptides. This TQ7WS product is reactive to alkynes, and useful for click chemistry.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 2112 | 1 mg | Price |
Physical properties
| Molecular weight | 984.12 |
| Solvent | DMSO |
Spectral properties
| Absorbance (nm) | 764 |
| Correction factor (260 nm) | 0.072 |
| Correction factor (280 nm) | 0.091 |
| Extinction coefficient (cm -1 M -1) | 140000 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on April 2, 2026

