Transfectamine™ 5000 Transfection Reagent
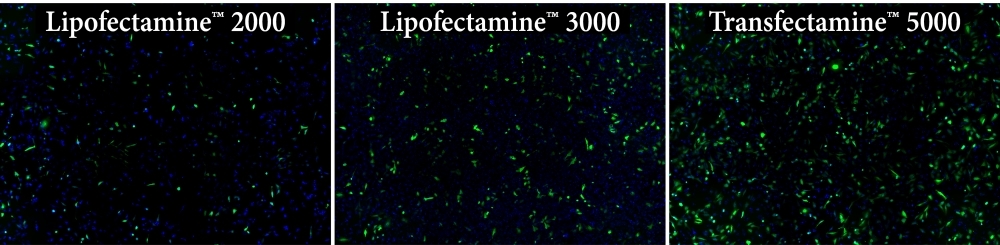
Transfectamine™ 5000 Transfection Reagent is a powerful and versatile transfection reagent for the introduction of nucleic acids into eukaryotic cells, or more specifically, into animal cells. It can effectively transfect a variety of payloads into a variety of adherent and suspension cell lines. It can be used for plasmid DNA transfection as well as siRNA- and shRNA-based gene knockdown experiments and gene expression studies. It offers consistently high transfection efficiency in a wide variety of adherent and suspension cell lines, including difficult-to-transfect cells. The low toxicity of Transfectamine™ 5000 also allowed higher viability of transfected cells. Transfectamine™ 5000 is easier to use compare to most other transfection reagents and does not require special medium.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 60019 | 50 uL | Price | |
| 60020 | 0.5 mL | Price | |
| 60021 | 1 mL | Price | |
| 60022 | 5 mL | Price |
Physical properties
| Solvent | Water |
Storage, safety and handling
| Intended use | Research Use Only (RUO) |
| Storage | Freeze (< -15 °C); Minimize light exposure |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 6, 2026
