SMCC and SMCC Plus™ Protein Crosslinkers
General Properties
Appearance: white crystal
Solvents: dimethylsulfoxide (DMSO) or dimethylformamide (DMF)
Note: SMCC Plus™ is soluble in water and many other aqueous buffers, in which it is less stable than in DMSO or DMF. Thus it is not recommended to make buffer stock solutions for repeated use. In addition, its solubility decreases with increasing salt concentration.
- SMCC NH2-reactive NHS ester moiety reacts rapidly with primary amine-containing molecule;
- SMCC HS-reactive maleimide moiety reacts with cysteine residues to yield specific conjugates;
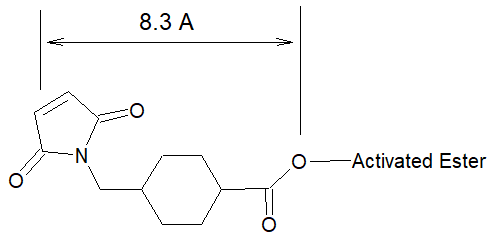
- Cyclohexane bridge confers added stability to the maleimide group making SMCC an ideal crosslinking agent for maleimide activation of proteins. Maleimide groups are stable at 4°C for 64 hours in 0.1 M sodium phosphate buffer (pH 7).
Biological Applications
Succinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (SMCC) is a non-cleavable and membrane-permeable crosslinker. It contains an amine-reactive N -hydroxysuccinimide (NHS ester) and a sulfhydryl-reactive maleimide group. The NHS ester moiety reacts with primary amines at pH 7-9 to form stable amide bonds. The maleimide moiety reacts with sulfhydryl groups at pH 6.5-7.5 to form stable thioether bonds. The maleimide groups of SMCC and SMCC Plus™ are unusually stable at pH up to 7.5 because of the cyclohexane bridge in the spacer arm.
SMCC Plus™, the water-soluble SMCC analog , is a heterobifunctional crosslinker that contains an activated ester and maleimide group that allow covalent conjugation of amine- and sulfhydryl-containing molecules. As SMCC, the activated ester moiety of SMCC Plus™ reacts with primary amines at pH 7-9 to form amide bonds, while its maleimide group reacts with sulfhydryl groups at pH 6.5-7.5 to form stable thioether bonds. The activated ester of SMCC Plus™ is more stable than the classic NHS ester of SMCC in aqueous solutions, but has similar reactivity to amines.
In aqueous solutions, the hydrolytic degradation of activated ester is a competing reaction whose rate increases with pH. The maleimide group is more stable than the activated ester group but will slowly hydrolyze and lose its reaction specificity for sulfhydryls at pH values >7.5. For these reasons, conjugations with this crosslinker are usually performed at pH 7.2-7.5, with the activated ester (amine-targeted) reacted either before or simultaneously with the maleimide (sulfhydryl-targeted) reaction.
SMCC is a reagent of choice for creating stable maleimide-activated carrier proteins that will spontaneously react with sulfhydryls. The cyclohexane ring in the spacer arm of this reagent decreases the rate of hydrolysis of the maleimide group compared to similar reagents that do not contain this ring. This feature enables proteins that have been maleimide-activated with SMCC Plus™ to be lyophilized and stored for later conjugation to a sulfhydryl-containing molecule. Many maleimide-activated protein products are produced in this manner. In this application, the NHS ester of SMCC is reacted with lysine residues on the carrier protein, converting them to reactive maleimides. These relatively stable maleimide-activated intermediates may be lyophilized and stored for later conjugation to a hapten.
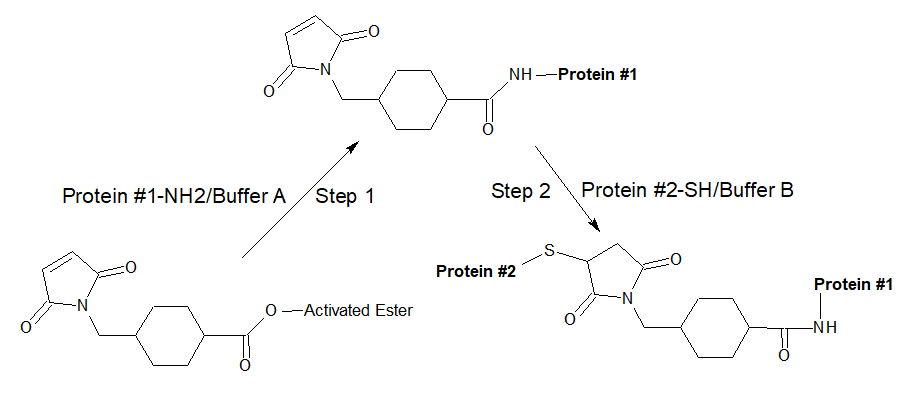
Our SMCC and SMCC Plus™ are used to prepare antibody-enzyme and hapten-carrier protein conjugates in a two-step reaction scheme as shown below. First, the amine-containing protein is reacted with a several-fold molar excess of the crosslinker, followed by the removal of excess (non-reacted) reagent by desalting or dialysis; finally, the sulfhydryl-containing molecule is added to react with the maleimide groups already attached to the first protein. The more detailed conjugation procedure is described below.
Procedure for Two-step Protein Cross-linking Using SMCC and SMCC Plus™
Generally, a 10- to 50-fold molar excess of SMCC or SMCC Plus™ over the amount of amine-containing protein results in sufficient maleimide activation to enable several sulfhydryl-containing proteins to be conjugated to each amine-containing protein. More dilute protein solutions require greater fold molar excess of reagent to achieve the same activation level. Empirical testing is necessary to determine optimal activation levels and final conjugation ratios for the intended application.
- Prepare conjugation buffer for amine-containing protein, Protein-NH2 (Buffer A): Phosphate buffered saline (PBS, pH 7.2) or other amine-free buffers with pH from 7.2 to 8.0 are OK to use for this purpose.
Note: Avoid buffers containing primary amines (e.g., Tris or glycine) and sulfhydryls during conjugation, because they will compete with the intended reaction. If necessary, dialyze or desalt samples into an appropriate buffer such as PBS. - Prepare conjugation buffer for sulfhydryl-containing protein, Protein-SH (Buffer B): Sulfhydryl-free buffers at pH from 6.5 to 7.5 are OK to use for this purpose.
Note 1: Avoid buffers containing sulfhydryls during conjugation, because they will compete with the intended reaction. If necessary, dialyze or desalt samples into an appropriate buffer such as PBS.
Note 2: The addition of 1-5 mM EDTA helps to chelate divalent metals, thereby reducing disulfide formation in the sulfhydryl-containing protein. - Prepare desalting column to separate modified protein from excess crosslinker and reaction by-products.
- Prepare amine-containing protein (Protein-NH2) and sulfhydryl-containing protein (Protein-SH) solutions in their Conjugation Buffer A and Buffer B.
- Prepare Protein 1-NH2-SMCC (or SMCC Plus™) conjugate: Add the appropriate amount of SMCC (or SMCC Plus™) to the amine-containing protein solution in Buffer A. The concentration of the Protein-NH2 determines SMCC (or SMCC Plus™) molar excess to use. The suggested volume of SMCC (or SMCC Plus™) molar excesses are as follows, but the best ratio has to be determined experimentally using the target proteins:
- Protein samples < 1 mg/mL use 40-80-fold molar excess.
- Protein samples of 1-4 mg/mL use 20-fold molar excess.
- Protein samples of 5-10 mg/mL use 5-to 10-fold molar excess.
- Incubate the above reaction mixture at room temperature for 30-60 minutes or at 4 °C for 2-4 hours.
Note 1: To stop the conjugation reaction before completion, add buffer containing reduced cysteine at a concentration several times greater than that of the sulfhydryls of Protein-SH.
Note 2: Conjugation efficiency can be estimated by electrophoresis separation and subsequent protein staining. - Remove excess SMCC (or SMCC Plus™) by using a desalting column equilibrated with Conjugation Buffer A.
- Combine and mix thiol-containing (Protein-SH) and desalted amine-containing protein (Protein-NH2) SMCC (or SMCC Plus™) conjugate in a molar ratio corresponding to that desired for the final conjugate and consistent with the relative number of sulfhydryl and activated amines that exist on the two proteins.
Note: Molecules to be reacted with the maleimide moiety must have free (reduced) sulfhydryls. For proteins, reduce disulfide bonds using 5 mM TCEP at room temperature for 30 minutes, followed by two passes through a suitable desalting column. Be aware that proteins (e.g., antibodies) may be inactivated by complete reduction of their disulfide bonds. Selective reduction of hinge-region disulfide bonds in IgG can be accomplished with 2-mercaptoethylamine•HCl (2-MEA). Sulfhydryls can be added to molecules using N-succinimidyl S-acetylthioacetate (SATA) or 2-iminothiolane•HCl (Traut's Reagent), which modify primary amines.
Storage Conditions
Store at 2°C to 8°C. Expiration date is one year from the date of receipt.
Note: SMCC (or SMCC Plus™) is moisture-sensitive. Store desiccated. Equilibrate vial to room temperature before opening to avoid moisture condensation. Dissolve needed amount of reagent and use immediately before hydrolysis occurs. Discard any unused reconstituted reagent. Do not store reagent in solution.
References
- Ishikawa, E., et al. (1983). Enzyme-labeling of antibodies. J. Immunoassay 4:209-327.
- Brinkley, M.A. (1992). A survey of methods for preparing protein conjugates with dyes, haptens and cross-linking reagents. Bioconjugate Chem. 3:2-13.
- Hashida, S., et al. (1984). More useful maleimide compounds for the conjugation of Fab to horseradish peroxidase through thiol groups in the hinge. J. Appl. Biochem. 6:56-63.
- Mattson, G., et al. (1993). A practical approach to cross-linking. Molecular Biology Reports 17:167-83.
- Partis, M.D., et al. (1983). Cross-linking of proteins by omega-maleimido alkanoyl N-hydroxysuccinimide esters. J. Protein. Chem. 2:263-77.
- Yoshitake, S., et al. (1982). Mild and efficient conjugation of rabbit Fab and horseradish peroxidase using a maleimide compound and its use for enzyme immunoassay. J. Biochem. 92:1413-24.
Original created on January 11, 2022, last updated on January 11, 2022
Tagged under: