Amplite® Fluorimetric Acetylcholine Assay Kit
Red Fluorescence
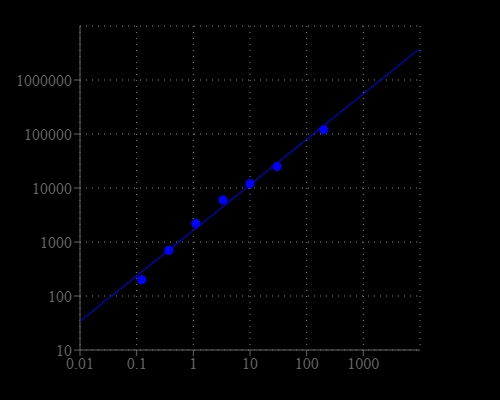
Acetylcholine and its metabolites are needed for three main physiological purposes: structural integrity and signaling roles for cell membranes, cholinergic neurotransmission (acetylcholine synthesis), and as a major source for methyl groups via its metabolite, trimethylglycine (betaine) that participates in the S-adenosylmethionine synthesis pathways. It plays an important role in the central nervous system as a precursor for acetylcholine and membrane phosphatidylcholine. This Amplite® Fluorimetric Acetylcholine Assay Kit provides one of the most sensitive methods for the quantifying acetylcholine. The kit uses Amplite® Red™ to quantify acetylcholine through choline oxidase-mediated enzyme coupling reactions. The fluorescence intensity of Amplite® Red™ is proportional to acetylcholine.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 11403 | 200 Tests | Price |
Spectral properties
| Excitation (nm) | 571 |
| Emission (nm) | 584 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12171501 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 540 nm |
| Emission | 590 nm |
| Cutoff | 570 nm |
| Recommended plate | Solid black |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 4, 2026

