trFluor™ Tb goat anti-rabbit IgG (H+L) *Cross Adsorbed*
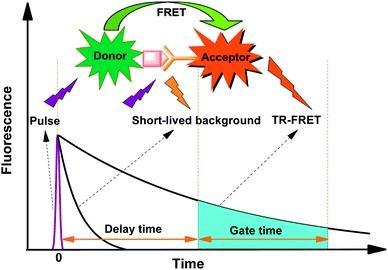
Many biological compounds present in cells, serum or other biological fluids are naturally fluorescent, and thus the use of conventional, prompt fluorophores leads to serious limitations in assay sensitivity due to the high background caused by the autofluorescence of the biological molecules to be assayed. The use of long-lived fluorophores combined with time-resolved detection (a delay between excitation and emission detection) minimizes prompt fluorescence interferences. Our trFluor™ Tb probes enable time-resolved fluorometry (TRF) for the assays that require high sensitivity. trFluor™ Tb probes have large Stokes shifts and extremely long emission half-lives when compared to more traditional fluorophores such as Alexa Fluor or cyanine dyes. Compared to the other TRF compounds, our trFluor™ Tb probes have relatively high stability, high emission yield and ability to be linked to biomolecules. This trFluor™ Tb goat anti-rabbit IgG (H+L) conjugate is commonly used as a second step reagent for indirect immunofluorescent staining, when used in conjunction with primary antibodies.
Spectrum
Product family
| Name | Excitation (nm) | Emission (nm) | Extinction coefficient (cm -1 M -1) | Correction Factor (260 nm) | Correction Factor (280 nm) |
| trFluor™ Eu goat anti-rabbit IgG (H+L) | 298 | 617 | 21000 | 0.911 | 0.777 |
| trFluor™ Eu goat anti-rabbit IgG (H+L) *Cross Adsorbed* | 298 | 617 | 21000 | 0.911 | 0.777 |
| trFluor™ Tb goat anti-mouse IgG (H+L) | 333 | 544 | - | 0.942 | 0.797 |
| trFluor™ Tb goat anti-mouse IgG (H+L) *Cross Adsorbed* | 333 | 544 | - | 0.942 | 0.797 |
References
View all 61 references: Citation Explorer
Development of a time-resolved fluorescence resonance energy transfer assay for cyclin-dependent kinase 4 and identification of its ATP-noncompetitive inhibitors
Authors: Lo MC, Ngo R, Dai K, Li C, Liang L, Lee J, Emkey R, Eksterowicz J, Ventura M, Young SW, Xiao SH.
Journal: Anal Biochem (2012): 368
Authors: Lo MC, Ngo R, Dai K, Li C, Liang L, Lee J, Emkey R, Eksterowicz J, Ventura M, Young SW, Xiao SH.
Journal: Anal Biochem (2012): 368
Time-Resolved Fluorescence Resonance Energy Transfer as a Versatile Tool in the Development of Homogeneous Cellular Kinase Assays
Authors: Saville L, Spais C, Mason JL, Albom MS, Murthy S, Meyer SL, Ator MA, Angeles TS, Husten J.
Journal: Assay Drug Dev Technol. (2012)
Authors: Saville L, Spais C, Mason JL, Albom MS, Murthy S, Meyer SL, Ator MA, Angeles TS, Husten J.
Journal: Assay Drug Dev Technol. (2012)
Oligomerization of the serotonin(1A) receptor in live cells: a time-resolved fluorescence anisotropy approach
Authors: Paila YD, Kombrabail M, Krishnamoorthy G, Chattopadhyay A.
Journal: J Phys Chem B (2011): 11439
Authors: Paila YD, Kombrabail M, Krishnamoorthy G, Chattopadhyay A.
Journal: J Phys Chem B (2011): 11439
A homogeneous single-label time-resolved fluorescence cAMP assay
Authors: Martikkala E, Rozw and owicz-Jansen A, Hanninen P, Petaja-Repo U, Harma H.
Journal: J Biomol Screen (2011): 356
Authors: Martikkala E, Rozw and owicz-Jansen A, Hanninen P, Petaja-Repo U, Harma H.
Journal: J Biomol Screen (2011): 356
Time-resolved fluorescence resonance energy transfer (TR-FRET) to analyze the disruption of EGFR/HER2 dimers: a new method to evaluate the efficiency of targeted therapy using monoclonal antibodies
Authors: Gaborit N, Larbouret C, Vallaghe J, Peyrusson F, Bascoul-Mollevi C, Crapez E, Azria D, Chardes T, Poul MA, Mathis G, Bazin H, Pelegrin A.
Journal: J Biol Chem (2011): 11337
Authors: Gaborit N, Larbouret C, Vallaghe J, Peyrusson F, Bascoul-Mollevi C, Crapez E, Azria D, Chardes T, Poul MA, Mathis G, Bazin H, Pelegrin A.
Journal: J Biol Chem (2011): 11337
Page updated on August 26, 2025