Transfection Reagents
Coaxing DNA or RNA past the hydrophobic cell membrane can prove challenging due to the hydrophilic properties of nucleic acids. To overcome this, scientists rely on a technique known as transfection to introduce exogenous nucleic acids (e.g., plasmid DNA, mRNA, siRNA, and oligonucleotides) into eukaryotic primary cells or cell lines in vitro or into carefully selected animal models in vivo. As a powerful analytical tool, transfection is widely used in gene expression analysis to study the overexpression or silencing of a gene of interest and its subsequent effect on protein expression and functionality, for the production of biologics, such as antibodies, or recombinant proteins, or recombinant viral particles, and in the development of gene-based therapies.
Stable and Transient Transfection
Depending upon the nature of the newly introduced genetic materials and their long-term effects on cells, transfected DNA can be classified as either stable or transient. In stable transfection, exogenous nucleic acids are incorporated into the nuclear genome or persist as an episomal plasmid, enabling transfected cells and their progeny to express the gene of interest permanently. Stable transfection is generally limited to DNA vectors, however, siRNA and miRNA delivered as short hairpin transcripts can also be stably transfected. Strategies for stable transfection are advantageous for prolonged experiments, such as recombinant protein production, gene regulation, gene therapy, and drug discovery.
In transient transfection, the genetic material does not get integrated into the host genome. As a result, the gene of interest is expressed only for a limited period of time, typically 24-96 hours, and is not transferred to daughter cells during cell division. Unlike stable transfection, transiently transfected genetic materials can be lost by environmental factors or diluted out during cell division. Transient transfection strategies are most efficient at transfecting supercoiled plasmid DNA but can also be used to transfect siRNAs, miRNAs, mRNAs, and proteins. Transient transfection is best-suited for short-term gene expression studies, such as gene knockdown or silencing using inhibitory RNAs and small-scale protein production.
Transfection Methods
While various approaches can be used to stably or transiently transfect genetic material, they broadly fall into three categories - physical, viral, and chemical methods. Of the three main types of transfection, chemical-mediated transfection is the most widely used in contemporary research for its ease and cost-effectiveness.
Physical-Mediated Transfection
Physical transfection methods, such as electroporation, microinjection, and biolistic particle delivery, rely on a diverse set of physical tools that use mechanical or electrical forces to deliver exogenous nucleic acids. Microinjection utilizes a micromanipulator and microscope to directly insert nucleic acids into the cytoplasm or nucleus. Although time-consuming and technically challenging, microinjection affords users the ability to control the amount and timing of injected materials delivered and is suitable for various cell types. Biolistic particle delivery, also called particle bombardment, uses a ballistic device to shoot nucleic acids coated with gold particles into cells at a high velocity. This approach may not be as precise as microinjection, but it offers a simple method to both transient and stable transfection, can be used on various tissue and cell types, including plants, and can co-deliver multiple plasmids with high frequencies of cotransfromation.
Of the various types of physical transfection, electroporation is the most widely used. In this method, an electrical field is applied to cells, which perturbs the cell membrane resulting in the formation of transient pores that facilitate the entry of exogenous material. Electroporation is applicable for transient and stable transfection of all cell types, and under optimal conditions, it can be adapted for high-throughput transfection. Although physical transfection is advantageous for its low cytotoxicity and ability to deliver genetic material rapidly and directly, these methods are relatively harsh on cells causing disruption to the cellular membrane and often results in substantial cell death.
Viral-Mediated Transfection
Viral-mediated transfection, also known as transduction, is widely used in clinical research for its high in vivo transfection efficiency and sustained gene expression without severely affecting cell viability. In contrast to chemical transfection methods, no transfection reagent is required. Instead, the viral vector infects the cells and delivers the genetic material directly to the nucleus. Common viral delivery systems include adenoviral, oncoretroviral, lentiviral, baculovirus, and vaccinia virus-based vectors. Even though viral-mediated transfection is highly effective, the major concerns associated with its usage are immunogenicity, cytotoxicity, and low packaging capacity.
Chemical-Mediated Transfection
Chemical-mediated transfection relies on electrostatic interactions to deliver exogenous nucleic acids into cells. In this method, electrostatic forces between the cationic charges on the lipid or polymer groups of transfection reagents and the negatively charged phosphates of nucleic acids cause the two substances to associate, resulting in positively charged transfection complexes. These cationic transfection complexes, in turn, interact with negatively-charged glycoproteins, such as heparan sulfate proteoglycans, expressed on the cell surface triggering the cellular uptake of exogenous nucleic acid via endocytosis or phagocytosis. Commonly used methods for chemical transfection include calcium phosphate, cationic polymer, and cationic lipid transfection (see Table 1).
Table 1. Overview of chemical transfection methods
| Chemical Method ▲ ▼ | Principle ▲ ▼ | Advantages ▲ ▼ | Disadvantages ▲ ▼ | Examples ▲ ▼ |
| Cationic polymer | Negatively charged nucleotide backbones form complexes with cationic polymers, which are then taken up by cells via endocytosis |
|
|
|
| Calcium phosphate co-precipitate | Nucleotides, calcium and phosphate buffer are combined to form a precipitate that is taken up by cells via endocytosis. |
|
|
|
| Cationic lipid | Negatively charged nucleotides form complexes with cationic lipids, which then pass through the cell membrane and release the nucleotides into they cytoplasm via endocytosis |
|
|
Transfectamine™ Transfection Reagents
Transfectamine™ transfection reagents, developed by AAT Bioquest, are cationic-lipid transfection reagents designed to deliver exogenous nucleic acids into eukaryotic cells. The superior transfection performance and versatility of Transfectamine™ transfection reagents enable users to effectively transfect various payloads into a wide variety of adherent and suspension cell lines, with exceptional efficiency, cell viability, and reproducibility.
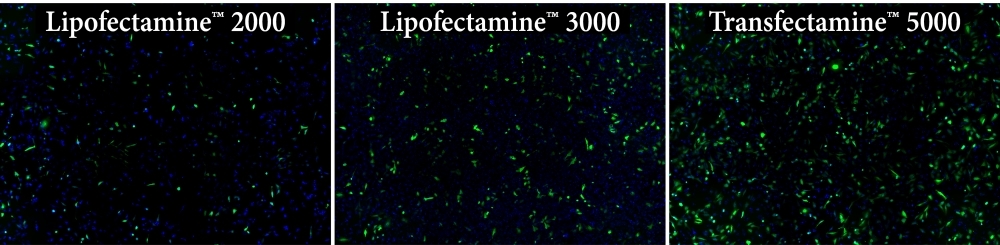
Transfection efficiency comparison in HeLa cells using Transfectamine™ 5000, Lipofectamine 2000, and Lipofectamine 3000 reagents. Each reagent was used to transfect HeLa cells in a 96-well format, and GFP expression was analyzed 24 hours post-transfection. Transfectamine™ 5000 transfection reagent provided higher GFP transfection efficiency compared to Lipofectamine 2000 and Lipofectamine 3000 reagents.
Table 2. Overview of Transfectamine™ transfection reagents
| Product ▲ ▼ | Cell Type ▲ ▼ | Adherent or Suspension ▲ ▼ | Payload ▲ ▼ | Delivery Method ▲ ▼ | Unit Size ▲ ▼ | Cat No. ▲ ▼ |
| Transfectamine™ 5000 |
|
|
|
|
| |
| Transfectamine™ mRNA |
|
|
|
|
|