Portelite™ Fluorimetric Lithium Ion Quantification Kit™ uses our robust lithium ion indicator dye, Lithiumighty™ 520, which exhibit great fluorescence intensity enhancement upon binding to Lithium ions. Lithiumighty™ 520 is the most robust lithium ion indicator with high selectivity. It enables the kit to be useful for the rapid determination of lithium concentrations in a variety of samples compared to the other commercial lithium ion assays. This nanodrop-based assay kit requires a small amount of sample, it is particularly suitable for the determination of lithium ion concentration in fields or on site. Quantifying lithium ions is important in various scientific fields and industries, including biochemistry, medicine, environmental analysis, and food science etc. The rapid and accurate determination of lithium ions is particularly important in the battery industry. There are several methods commonly used to quantify lithium ions, including flame photometry, ion-selective electrodes (ISE), atomic absorption spectroscopy and fluorescence spectrophotometry. Flame photometry and atomic absorption spectroscopy require the inflammation of the samples. They are tedious to use and require expensive and sophisticated instrumentation. Ion-selective electrodes require large volumes of samples and often have low selectivity. Among all the methods, fluorescence spectrophotometry is the most convenient method for quantifying lithium ions. Fluorescence spectrophotometry involves complexing lithium ions with specific reagents and measuring the resulting fluorescence changes. However, there is still a lack of a fluorescence-based lithium ion assay kit in the commercial market due to the absence of a robust fluorescence lithium ion indicator. For the first time, Lithiumighty™ 520 filled this gap. It is the best fluorescent lithium ion indicator for rapidly determining lithium ion concentration in combination with a fluorescence device such as a fluorescence Nanodrop spectrophotometer or a fluorescence microplate reader.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 21353 | 50 Tests | Price |
| Excitation (nm) | 491 |
| Emission (nm) | 513 |
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
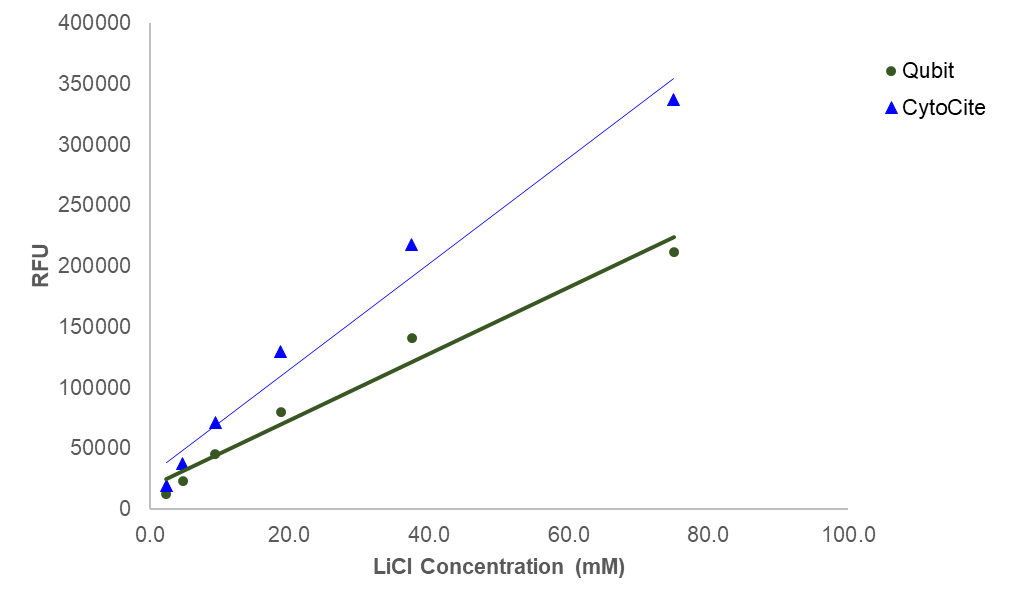
| Qubit Fluorometer | |
| Excitation | 480 nm |
| Emission | 530 nm |
| Instrument specification(s) | 0.2 mL PCR tube |
| CytoCite Fluorometer | |
| Excitation | 480 nm |
| Emission | 530 nm |
| Instrument specification(s) | 0.2 mL PCR tube |
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |

