CytoTell® Red 590
CytoTell® Red 590 is a red fluorescent cell proliferation dye for monitoring cell division and long-term tracking of live cells in multicolor flow cytometry and fluorescence microscopy applications.
- Reduced cytotoxicity: Provides lower cellular toxicity for long-term live cell proliferation studies
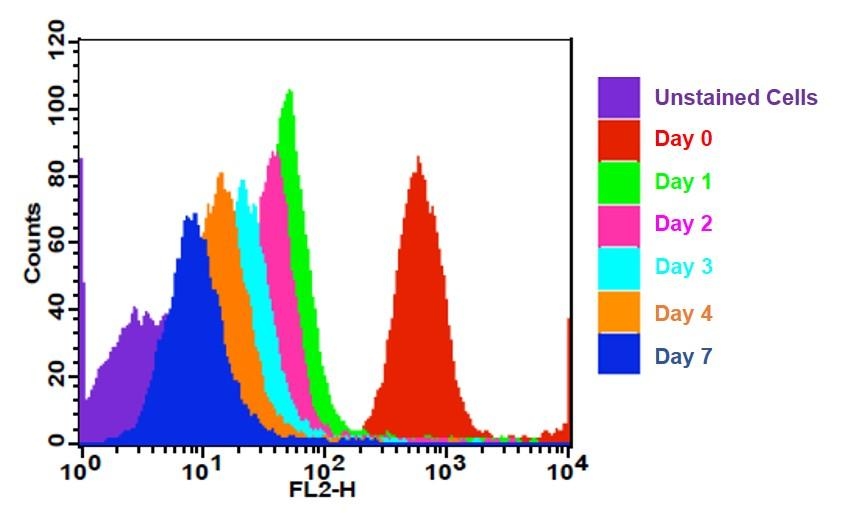
- Enhanced proliferation tracking: Enables monitoring of successive cell generations through progressive fluorescence dilution
- 561 nm laser compatible: Optimized for excitation with yellow laser-based flow cytometers and imaging systems
- Distinct spectral profile: Provides spectral separation from GFP and FITC for multicolor cell analysis applications
- Broad application compatibility: Suitable for flow cytometry, fluorescence microscopy, intracellular staining workflows, and multicolor cell analysis applications

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 22261 | 500 Tests | Price | |
| 22262 | 2x500 Tests | Price |
Physical properties
| Molecular weight | 619.53 |
| Solvent | DMSO |
Spectral properties
| Excitation (nm) | 562 |
| Emission (nm) | 587 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
Instrument settings
| Flow cytometer | |
| Excitation | 488 nm or 561nm laser |
| Emission | 610/20 nm filter |
| Instrument specification(s) | PE-Texas Red channel |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 6, 2026

