ReadiUse™ Yeast Cell Lysis Buffer *5X*
Ordering information
| Price | |
| Catalog Number | |
| Unit Size | |
| Quantity |
Additional ordering information
| Telephone | 1-800-990-8053 |
| Fax | 1-800-609-2943 |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Shipping | Standard overnight for United States, inquire for international |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Refrigerated (2-8 °C) |
| UNSPSC | 12352200 |
Related products
| Overview |
ReadiUse™ Yeast Cell Lysis Buffer is a biochemical reagent designed for efficient yeast cell lysis and protein solubilization in laboratory applications. Engineered to simplify the extraction of cell components, including proteins, lipids, nucleic acids, reporter molecules, and small biomolecules, this buffer employs a detergent-based lysis procedure for the gentle breakdown of yeast cell membranes. It releases functionally active and solubilized proteins without the need for abrasive methods like glass beads or mechanical treatments with strong reducing agents. With a mild detergent formulation, ReadiUse™ Yeast Cell Lysis Buffer minimizes the reliance on harsh chemical treatments, extreme pH adjustments, or temperature variations, contributing to the preservation of extracted protein integrity. Versatile in its efficacy across various organisms, including Bacillus subtilis, Gram-positive, and Gram-negative bacteria, ReadiUse™ Yeast Cell Lysis Buffer eliminates the need for external disruptive factors, simplifying the cell lysis process. Its applications span molecular biology research, protein purification, analysis, and biomolecular studies, offering a reliable solution for scientific experimentation.
Example protocol
AT A GLANCE
Important
Expiration date is 6 months from the date of receipt.
PREPARATION OF WORKING SOLUTION
ReadiUse™ Yeast Cell Lysis Buffer Working Solution (1X)
Add 1 mL of ReadiUse™ Yeast Cell Lysis Buffer (5X) into 4 mL of Mili-Q water and mix well.
SAMPLE EXPERIMENTAL PROTOCOL
Following is our recommended protocol for loading AM esters into live cells. This protocol only provides a guideline and should be modified according to your specific needs.
Prepare yeast cell samples by collecting yeast cells via centrifugation (10,000 g, 0°C, 15 minutes). Use about 100 to 10 million cells/mL of ReadiUse™ Yeast Cell Lysis Buffer working solution (1X), and keep the treated solution at room temperature for 15 minutes.
Centrifuge at 2500 rpm for 5 minutes, and use the supernatant for further biochemical analysis.
Images
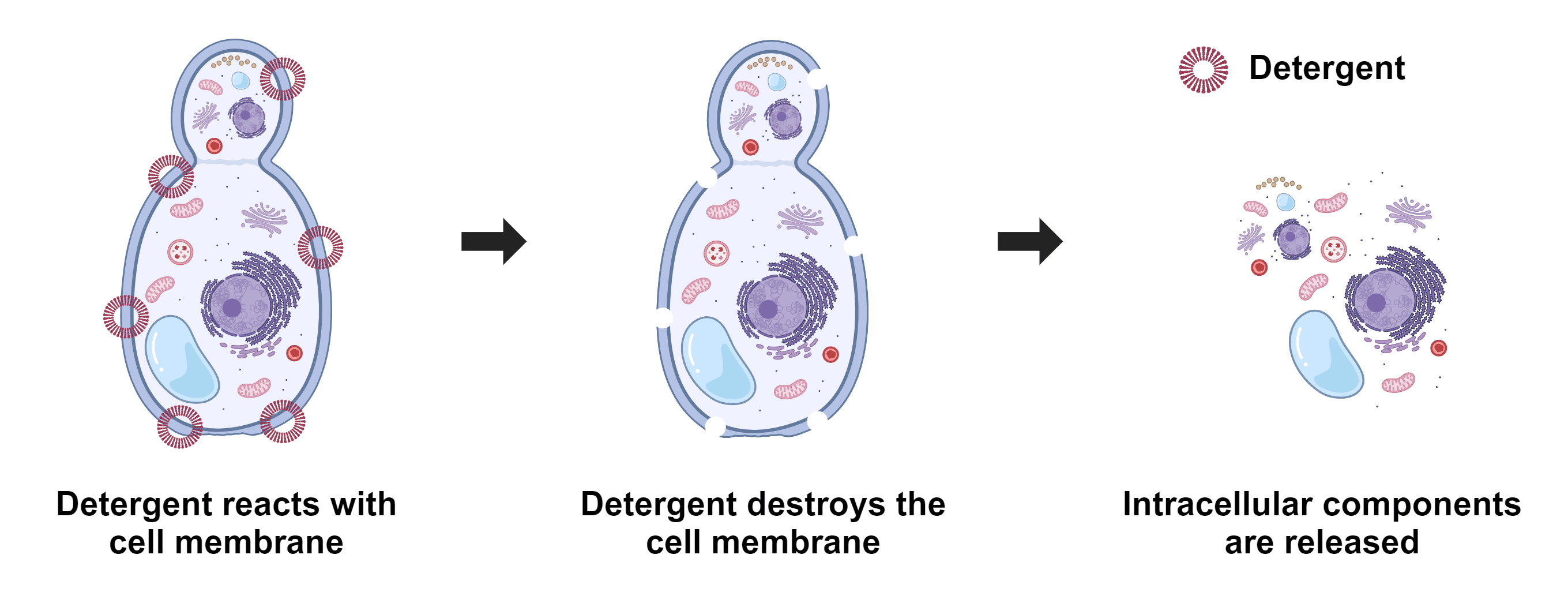
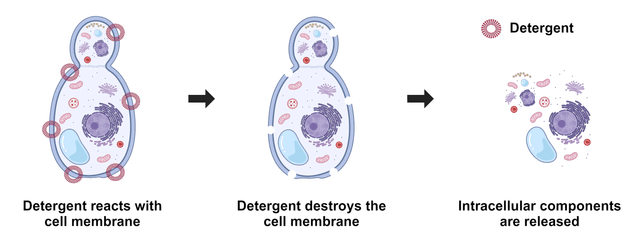
Figure 1. Yeast cell lysis refers to the breaking down of yeast cells, and it is often used to analyze specific cellular compositions such as proteins, lipids, nucleic acids, reporter molecules, cell signal molecules, and other small biomolecules. Depending upon the detergents used, either all or some membranes are lysed. ReadiUse™ reagents require minimal hands-on time. This yeast cell lysis buffer just requires a simple 5-fold dilution.
References
View all 19 references: Citation Explorer
Evaluation of Physicochemical and Microbial Properties of Extracts from Wine Lees Waste of Matelica's Verdicchio and Their Applications in Novel Cosmetic Products.
Authors: Di Nicolantonio, Lucrezia and Ferrati, Marta and Cristino, Maria and Peregrina, Dolores Vargas and Zannotti, Marco and Vitali, Luca Agostino and Ciancia, Sonia Ilaria and Giovannetti, Rita and Ferraro, Stefano and Zara, Susi and Di Valerio, Valentina and Cataldi, Amelia and Gigliobianco, Maria Rosa and Censi, Roberta and Di Martino, Piera
Journal: Antioxidants (Basel, Switzerland) (2023)
Authors: Di Nicolantonio, Lucrezia and Ferrati, Marta and Cristino, Maria and Peregrina, Dolores Vargas and Zannotti, Marco and Vitali, Luca Agostino and Ciancia, Sonia Ilaria and Giovannetti, Rita and Ferraro, Stefano and Zara, Susi and Di Valerio, Valentina and Cataldi, Amelia and Gigliobianco, Maria Rosa and Censi, Roberta and Di Martino, Piera
Journal: Antioxidants (Basel, Switzerland) (2023)
Cloning and Overexpressing Membrane Proteins Using Pichia pastoris (Komagataella phaffii).
Authors: Schwob, Magali and Kugler, Valérie and Wagner, Renaud
Journal: Current protocols (2023): e936
Authors: Schwob, Magali and Kugler, Valérie and Wagner, Renaud
Journal: Current protocols (2023): e936
Reliable Approach for Pure Yeast Cell Wall Protein Isolation from Saccharomyces cerevisiae Yeast Cells.
Authors: Yammine, Marie and Bray, Fabrice and Flament, Stéphanie and Picavet, Antoine and Lacroix, Jean-Marie and Poilpré, Emmanuel and Mouly, Isabelle and Rolando, Christian
Journal: ACS omega (2022): 29702-29713
Authors: Yammine, Marie and Bray, Fabrice and Flament, Stéphanie and Picavet, Antoine and Lacroix, Jean-Marie and Poilpré, Emmanuel and Mouly, Isabelle and Rolando, Christian
Journal: ACS omega (2022): 29702-29713
Optimised Production and Extraction of Astaxanthin from the Yeast Xanthophyllomyces dendrorhous.
Authors: Harith, Zuharlida Tuan and de Andrade Lima, Micael and Charalampopoulos, Dimitris and Chatzifragkou, Afroditi
Journal: Microorganisms (2020)
Authors: Harith, Zuharlida Tuan and de Andrade Lima, Micael and Charalampopoulos, Dimitris and Chatzifragkou, Afroditi
Journal: Microorganisms (2020)
Effects of farnesol and lyticase on the formation of Candida albicans biofilm.
Authors: Sachivkina, Nadezhda and Lenchenko, Ekaterina and Blumenkrants, Dmitri and Ibragimova, Alfia and Bazarkina, Olga
Journal: Veterinary world (2020): 1030-1036
Authors: Sachivkina, Nadezhda and Lenchenko, Ekaterina and Blumenkrants, Dmitri and Ibragimova, Alfia and Bazarkina, Olga
Journal: Veterinary world (2020): 1030-1036
Comparison of the efficiency of different cell lysis methods and different commercial methods for RNA extraction from Candida albicans stored in RNAlater.
Authors: Rodríguez, Antonio and Vaneechoutte, Mario
Journal: BMC microbiology (2019): 94
Authors: Rodríguez, Antonio and Vaneechoutte, Mario
Journal: BMC microbiology (2019): 94
Cell lysis induced by membrane-damaging detergent saponins from Quillaja saponaria.
Authors: Berlowska, Joanna and Dudkiewicz, Marta and Kregiel, Dorota and Czyzowska, Agata and Witonska, Izabela
Journal: Enzyme and microbial technology (2015): 44-8
Authors: Berlowska, Joanna and Dudkiewicz, Marta and Kregiel, Dorota and Czyzowska, Agata and Witonska, Izabela
Journal: Enzyme and microbial technology (2015): 44-8
Yeast cell lysis enhances dimethyl trisulfide formation in sake.
Authors: Nishibori, Nahoko and Sasaki, Kei and Okimori, Yuta and Kanai, Muneyoshi and Isogai, Atsuko and Yamada, Osamu and Fujii, Tsutomu and Goto-Yamamoto, Nami
Journal: Journal of bioscience and bioengineering (2014): 526-8
Authors: Nishibori, Nahoko and Sasaki, Kei and Okimori, Yuta and Kanai, Muneyoshi and Isogai, Atsuko and Yamada, Osamu and Fujii, Tsutomu and Goto-Yamamoto, Nami
Journal: Journal of bioscience and bioengineering (2014): 526-8
Statistical analysis of sake-preparation conditions and dimethyl trisulfide formation.
Authors: Sasaki, Kei and Nishibori, Nahoko and Kanai, Muneyoshi and Isogai, Atsuko and Yamada, Osamu and Goto-Yamamoto, Nami and Fujii, Tsutomu
Journal: Journal of bioscience and bioengineering (2014): 166-71
Authors: Sasaki, Kei and Nishibori, Nahoko and Kanai, Muneyoshi and Isogai, Atsuko and Yamada, Osamu and Goto-Yamamoto, Nami and Fujii, Tsutomu
Journal: Journal of bioscience and bioengineering (2014): 166-71
Effects of yeast-originating polymeric compounds on ethanol pervaporation.
Authors: Gaykawad, S S and van der Wielen, L A M and Straathof, A J J
Journal: Bioresource technology (2012): 9-14
Authors: Gaykawad, S S and van der Wielen, L A M and Straathof, A J J
Journal: Bioresource technology (2012): 9-14