Amplite® Fluorimetric Glutamate Oxidase Assay Kit
Red Fluorescence
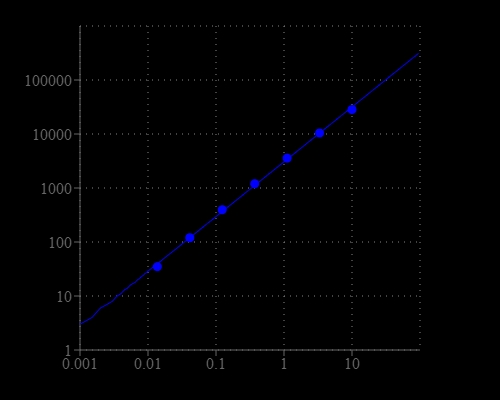
Glutamate oxidase belongs to the family of oxidoreductases, specifically those acting on the CH-NH2 group of donors with oxygen as an acceptor. It is an enzyme that specifically catalyzes the oxidative deamination of L-glutamate in the presence of water and oxygen with the formation of o-ketoglutarate, ammonia, and hydrogen peroxide. The Amplite® Fluorimetric Glutamate Oxidase Assay Kit provides a quick and ultrasensitive method for the measurement of glutamate oxidase in solution and in cell lysates. In the assay, L-glutamic acid is oxidized to µ-ketoglutarate, NH3 and H2O2 by glutamate oxidase. L-Alanine and L-glutamate-pyruvate transaminase are included in the reaction, resulting in multiple cycles of the initial reaction, thus significantly amplifying the production of H2O2. The kit uses our Amplite® Red substrate which enables a dual recordable mode. The fluorescent signal can be easily read by either a fluorescence microplate reader or an absorbance microplate reader. With the Amplite® Glutamate Oxidase Assay kit, we have detected as little as 40 uU/mL glutamate oxidase in a 100 µL reaction volume. It can be performed in a convenient 96-well or 384-well microtiter-plate format and easily adapted to automation without a separation step.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 11302 | 200 Tests | Price |
Spectral properties
| Excitation (nm) | 571 |
| Emission (nm) | 584 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12171501 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 540 nm |
| Emission | 590 nm |
| Cutoff | 570 nm |
| Recommended plate | Solid black |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 5, 2026

