Nuclear Blue™ LCS1
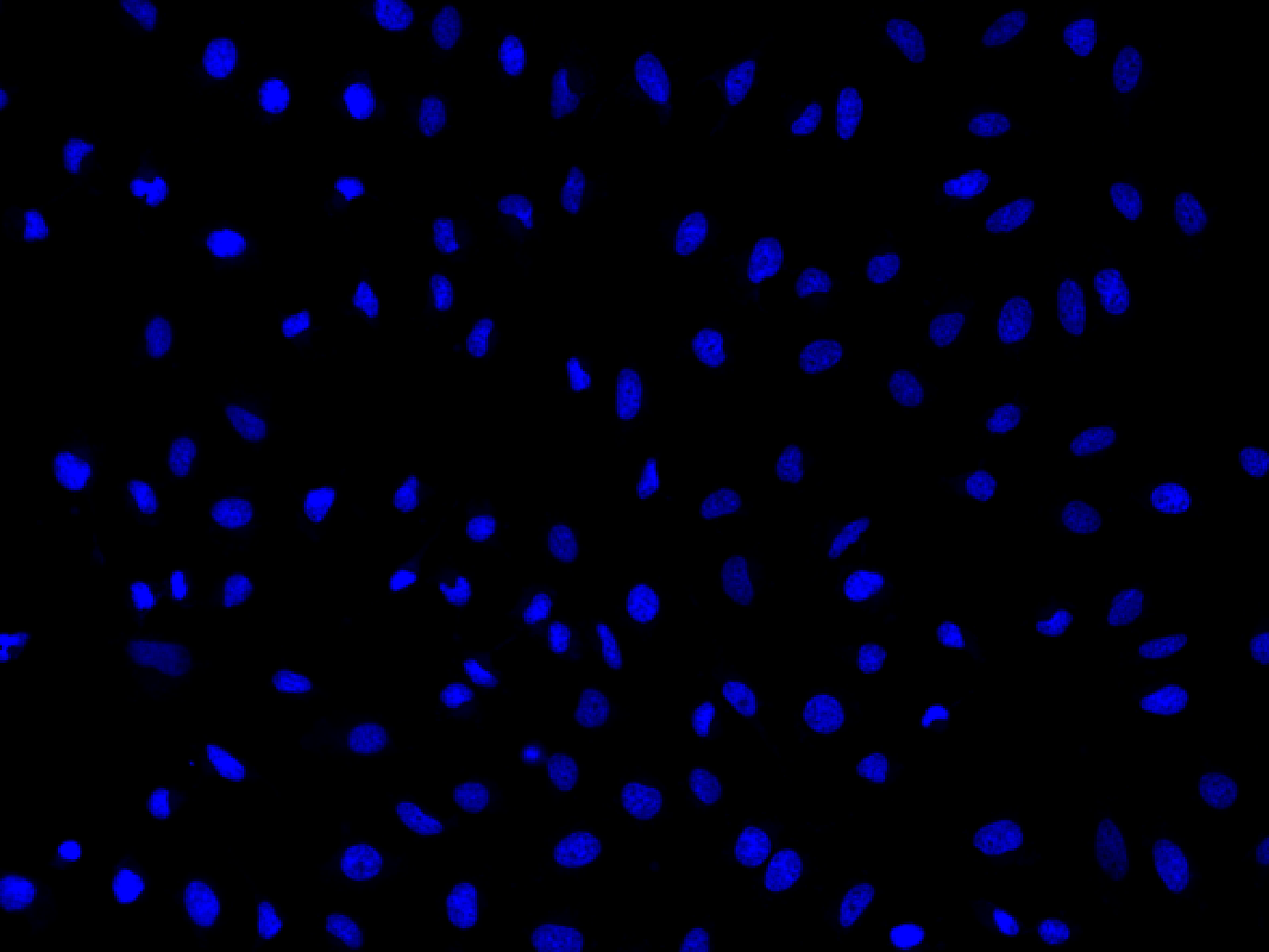
Our Nuclear Blue™ LCS1 is a fluorogenic, DNA-selective, and cell-permeant dye for analyzing DNA content in live cells. The Nuclear Blue™ LCS1 has its blue fluorescence significantly enhanced upon binding to DNA. It can be used in fluorescence imaging, microplate, and flow cytometry applications. This DNA-binding dye might be used for the multicolor analysis of live cells with the DAPI filter sets. For example, Nuclear Blue™LCS1 can be used with GFP cell lines.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 17559 | 0.5 mL | Price |
Physical properties
| Molecular weight | 822.69 |
| Solvent | DMSO |
Spectral properties
| Excitation (nm) | 353 |
| Emission (nm) | 456 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | T |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 41116134 |
Instrument settings
| Fluorescence microscope | |
| Excitation | DAPI Filter |
| Emission | DAPI Filter |
| Recommended plate | Black wall/clear bottom |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 5, 2026

