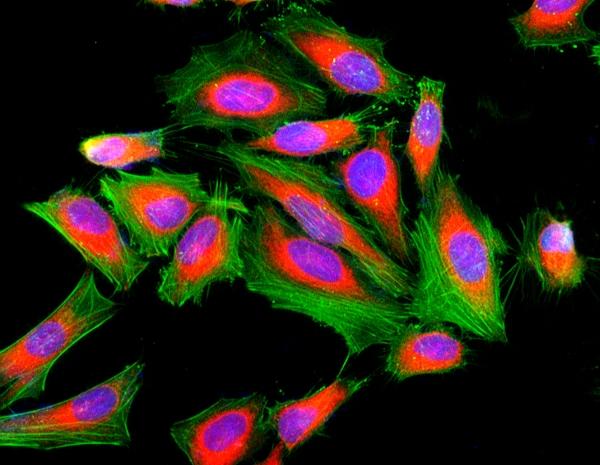
Cell Navigator® F-Actin Labeling Kit *Green Fluorescence*
Ordering information
| Price | |
| Catalog Number | |
| Unit Size | |
| Quantity |
Additional ordering information
| Telephone | 1-800-990-8053 |
| Fax | 1-800-609-2943 |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Shipping | Standard overnight for United States, inquire for international |
Spectral properties
| Correction Factor (260 nm) | 0.21 |
| Correction Factor (280 nm) | 0.11 |
| Extinction coefficient (cm -1 M -1) | 750001 |
| Excitation (nm) | 491 |
| Emission (nm) | 516 |
| Quantum yield | 0.91 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Alternative formats
| Cell Navigator® F-Actin Labeling Kit *Blue Fluorescence* |
| Cell Navigator® F-Actin Labeling Kit *Orange Fluorescence* |
| Cell Navigator® F-Actin Labeling Kit *Red Fluorescence* |
Related products
| Overview |
See also: Mitochondria, Cytoskeleton Structure & Analysis
Correction Factor (260 nm) 0.21 | Correction Factor (280 nm) 0.11 | Extinction coefficient (cm -1 M -1) 750001 | Excitation (nm) 491 | Emission (nm) 516 | Quantum yield 0.91 |
Our Cell Navigator® fluorescence imaging kits are a set of fluorescence imaging tools for labeling sub-cellular organelles such as membranes, lysosomes, mitochondria and nuclei etc. The selective labeling of live cell compartments provides a powerful method for studying cellular events in a spatial and temporal context. This particular kit is designed to label F-actins of fixed cells in green fluorescence. The kit uses a green fluorescent phalloidin conjugate that is selectively bound to F-actins. This green fluorescent phalloidin conjugate is a high-affinity probe for F-actins with much higher photostability than the fluorescein-phalloidin conjugates. Used at nanomolar concentrations, phallotoxins are convenient probes for labeling, identifying and quantitating F-actins in formaldehyde-fixed and permeabilized tissue sections, cell cultures or cell-free experiments. The labeling protocol is robust, requiring minimal hands-on time. The kit provides all the essential components with an optimized staining protocol.
Platform
Fluorescence microscope
| Excitation | FITC filter |
| Emission | FITC filter |
| Recommended plate | Black wall/clear bottom |
Components
Example protocol
AT A GLANCE
Protocol summary
- Prepare samples (microplate wells)
- Remove the liquid from the plate
- Add 100 µL/well of iFluor™ 488-Phalloidin working solution
- Stain the cells at RT for 15 to 60 minutes
- Wash the cells
- Examine the specimen under fluorescence microscope at Ex/Em = 490/520 nm (FITC filter set)
Important notes
Thaw all the components at room temperature before starting the experiment.
PREPARATION OF WORKING SOLUTION
Add 10 μL of iFluor™ 488-Phalloidin (Component A) to 10 mL of Labeling Buffer (Component B) to make 1X iFluor™ 488-Phalloidin working solution. Protect from light. Note: Different cell types might be stained differently. The concentration of iFluor™ 488-Phalloidin working solution should be prepared accordingly.
For guidelines on cell sample preparation, please visit
https://www.aatbio.com/resources/guides/cell-sample-preparation.html
SAMPLE EXPERIMENTAL PROTOCOL
- Perform formaldehyde fixation. Incubate the cells with 3.0% – 4.0% formaldehyde in PBS at room temperature for 10 – 30 minutes. Note: Avoid any methanol containing fixatives since methanol can disrupt actin during the fixation process. The preferred fixative is methanol-free formaldehyde.
- Rinse the fixed cells 2 – 3 times in PBS.
- Optional: Add 0.1% Triton X-100 in PBS into fixed cells for 3 to 5 minutes to increase permeability. Rinse the cells 2 – 3 times in PBS.
- Add 100 µL/well (96-well plate) of iFluor™ 488-Phalloidin working solution into the fixed cells.
- Stain the cells at room temperature for 15 to 60 minutes.
- Rinse cells gently with PBS 2 to 3 times to remove excess dye before plate sealing.
- Image cells using a fluorescence microscope with FITC filter set (Ex/Em = 490/520 nm).
Spectrum
Open in Advanced Spectrum Viewer
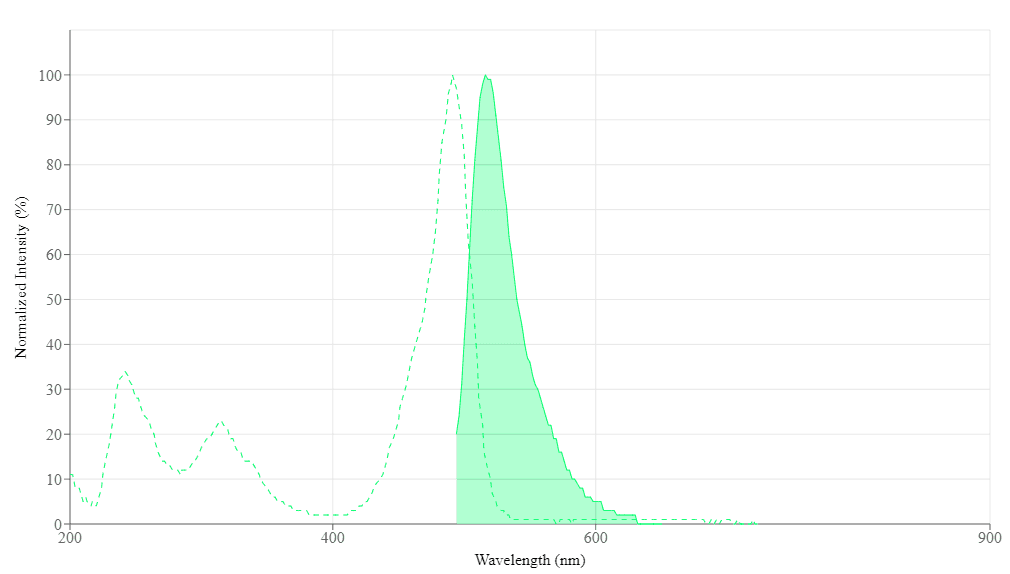

Spectral properties
| Correction Factor (260 nm) | 0.21 |
| Correction Factor (280 nm) | 0.11 |
| Extinction coefficient (cm -1 M -1) | 750001 |
| Excitation (nm) | 491 |
| Emission (nm) | 516 |
| Quantum yield | 0.91 |
Product Family
| Name | Excitation (nm) | Emission (nm) | Extinction coefficient (cm -1 M -1) | Quantum yield | Correction Factor (260 nm) | Correction Factor (280 nm) |
| Cell Navigator® F-Actin Labeling Kit *Blue Fluorescence* | 345 | 450 | 200001 | 0.951 | 0.83 | 0.23 |
| Cell Navigator® F-Actin Labeling Kit *Orange Fluorescence* | 541 | 557 | 1000001 | 0.671 | 0.25 | 0.15 |
| Cell Navigator® F-Actin Labeling Kit *Red Fluorescence* | 587 | 603 | 2000001 | 0.531 | 0.05 | 0.04 |
Images
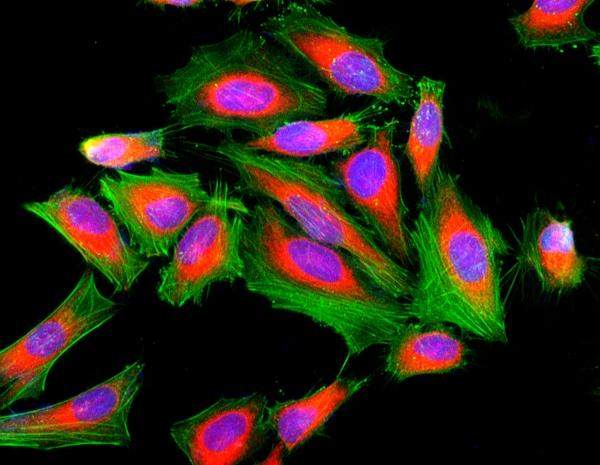
Figure 1. Fluorescence image of HeLa cells fixed with 4% formaldehyde then stained with Cell Navigator® F-Actin Labeling Kit *Green Fluorescence* in a Costar black 96-well plate. Cell were labeled with iFluor® 488-Phalloidin (Cat#22261, Green) and nuclei stain DAPI (Cat#17507, Blue), respectively. Cell endoplasmic reticulum (ER) was stained with ER Red™ (Cat#22636, Red) before fixation.
Citations
View all 42 citations: Citation Explorer
Temperature-Controlled Screening of Catechol Groups in Poly (N-Isopropylacrylamide-co-Dopamine Methacrylamide) for Cell Detachment
Authors: Yang, Liuxin and Ren, Pengfei and Wei, Dandan and Liang, Min and Xu, Li and Tao, Yinghua and Jiao, Guanhua and Zhang, Tianzhu and Zhang, Qianli
Journal: ACS Applied Polymer Materials (2024)
Authors: Yang, Liuxin and Ren, Pengfei and Wei, Dandan and Liang, Min and Xu, Li and Tao, Yinghua and Jiao, Guanhua and Zhang, Tianzhu and Zhang, Qianli
Journal: ACS Applied Polymer Materials (2024)
Reproduction of Entomopathogenic Nematodes for Use in Pest Control
Authors: Abd-Elgawad, Mahfouz MM
Journal: (2024): 351--382
Authors: Abd-Elgawad, Mahfouz MM
Journal: (2024): 351--382
TRPA1 and TPRV1 Ion Channels Are Required for Contact Lens-Induced Corneal Parainflammation and Can Modulate Levels of Resident Corneal Immune Cells
Authors: Datta, Ananya and Lee, Ji Hyun and Flandrin, Orneika and Horneman, Hart and Lee, Justin and Metruccio, Matteo ME and Bautista, Diana and Evans, David J and Fleiszig, Suzanne MJ
Journal: Investigative Ophthalmology \& Visual Science (2023): 21--21
Authors: Datta, Ananya and Lee, Ji Hyun and Flandrin, Orneika and Horneman, Hart and Lee, Justin and Metruccio, Matteo ME and Bautista, Diana and Evans, David J and Fleiszig, Suzanne MJ
Journal: Investigative Ophthalmology \& Visual Science (2023): 21--21
Modification of adipose mesenchymal stem cells-derived small extracellular vesicles with fibrin-targeting peptide CREKA for enhanced bone repair
Authors: Wu, Qi and Fu, Xiaoling and Li, Xian and Li, Jing and Han, Weiju and Wang, Yingjun
Journal: Bioactive Materials (2023): 208--220
Authors: Wu, Qi and Fu, Xiaoling and Li, Xian and Li, Jing and Han, Weiju and Wang, Yingjun
Journal: Bioactive Materials (2023): 208--220
Enhancing osteoinduction and bone regeneration of biphasic calcium phosphate scaffold thought modulating the balance between pro-osteogenesis and anti-osteoclastogenesis by zinc doping
Authors: Lu, T and Yuan, X and Zhang, L and He, F and Wang, X and Ye, J
Journal: Materials Today Chemistry (2023): 101410
Authors: Lu, T and Yuan, X and Zhang, L and He, F and Wang, X and Ye, J
Journal: Materials Today Chemistry (2023): 101410
Microfluidic Chip-Based Modeling of Three-Dimensional Intestine--Vessel--Liver Interactions in Fluorotelomer Alcohol Biotransformation
Authors: Xu, Ning and Lin, Haifeng and Lin, Jin-Ming and Cheng, Jie and Wang, Peilong and Lin, Ling
Journal: Analytical Chemistry (2023)
Authors: Xu, Ning and Lin, Haifeng and Lin, Jin-Ming and Cheng, Jie and Wang, Peilong and Lin, Ling
Journal: Analytical Chemistry (2023)
Digital light processing-bioprinted poly-NAGA-GelMA-based hydrogel lenticule for precise refractive errors correction
Authors: Jia, Shuo and Yang, Jirong and Lau, Aaron Dzi-Shing and Chen, Fushun and Bu, Yashan and Cai, Erlong and Wang, Huogang and Chieng, Herng-Ee and Sun, Tianhao and Zhiyong, Zhou and others,
Journal: Biofabrication (2023)
Authors: Jia, Shuo and Yang, Jirong and Lau, Aaron Dzi-Shing and Chen, Fushun and Bu, Yashan and Cai, Erlong and Wang, Huogang and Chieng, Herng-Ee and Sun, Tianhao and Zhiyong, Zhou and others,
Journal: Biofabrication (2023)
Honeycomb Biosilica in Sponges: From Understanding Principles of Unique Hierarchical Organization to Assessing Biomimetic Potential
Authors: Voronkina, Alona and Romanczuk-Ruszuk, Eliza and Przekop, Robert E and Lipowicz, Pawel and Gabriel, Ewa and Heimler, Korbinian and Rogoll, Anika and Vogt, Carla and Frydrych, Milosz and Wienclaw, Pawel and others,
Journal: Biomimetics (2023): 234
Authors: Voronkina, Alona and Romanczuk-Ruszuk, Eliza and Przekop, Robert E and Lipowicz, Pawel and Gabriel, Ewa and Heimler, Korbinian and Rogoll, Anika and Vogt, Carla and Frydrych, Milosz and Wienclaw, Pawel and others,
Journal: Biomimetics (2023): 234
Adjusting physicochemical and cytological properties of biphasic calcium phosphate by magnesium substitution: An in vitro study
Authors: Lu, Teliang and Miao, Yali and Yuan, Xinyuan and Zhang, Yu and Ye, Jiandong
Journal: Ceramics International (2023)
Authors: Lu, Teliang and Miao, Yali and Yuan, Xinyuan and Zhang, Yu and Ye, Jiandong
Journal: Ceramics International (2023)
CircRPAP2 regulates the alternative splicing of PTK2 by binding to SRSF1 in breast cancer
Authors: Yu, Yunhe and Fang, Lin
Journal: Cell death discovery (2022): 1--12
Authors: Yu, Yunhe and Fang, Lin
Journal: Cell death discovery (2022): 1--12
References
View all 42 references: Citation Explorer
Velocity distributions of single F-actin trajectories from a fluorescence image series using trajectory reconstruction and optical flow mapping
Authors: von Wegner F, Ober T, Weber C, Schurmann S, Winter R, Friedrich O, Fink RH, Vogel M.
Journal: J Biomed Opt (2008): 54018
Authors: von Wegner F, Ober T, Weber C, Schurmann S, Winter R, Friedrich O, Fink RH, Vogel M.
Journal: J Biomed Opt (2008): 54018
Visualization of F-actin and G-actin equilibrium using fluorescence resonance energy transfer (FRET) in cultured cells and neurons in slices
Authors: Okamoto K, Hayashi Y.
Journal: Nat Protoc (2006): 911
Authors: Okamoto K, Hayashi Y.
Journal: Nat Protoc (2006): 911
The effect of F-actin on the relay helix position of myosin II, as revealed by tryptophan fluorescence, and its implications for mechanochemical coupling
Authors: Conibear PB, Malnasi-Csizmadia A, Bagshaw CR.
Journal: Biochemistry (2004): 15404
Authors: Conibear PB, Malnasi-Csizmadia A, Bagshaw CR.
Journal: Biochemistry (2004): 15404
Analysis of models of F-actin using fluorescence resonance energy transfer spectroscopy
Authors: Moens PD, dos Remedios CG.
Journal: Results Probl Cell Differ (2001): 59
Authors: Moens PD, dos Remedios CG.
Journal: Results Probl Cell Differ (2001): 59
Fluorescence studies of the carboxyl-terminal domain of smooth muscle calponin effects of F-actin and salts
Authors: Bartegi A, Roustan C, Kassab R, Fattoum A.
Journal: Eur J Biochem (1999): 335
Authors: Bartegi A, Roustan C, Kassab R, Fattoum A.
Journal: Eur J Biochem (1999): 335
Microquantification of cellular and in vitro F-actin by rhodamine phalloidin fluorescence enhancement
Authors: Katanaev VL, Wymann MP.
Journal: Anal Biochem (1998): 185
Authors: Katanaev VL, Wymann MP.
Journal: Anal Biochem (1998): 185
A conformational change in F-actin when myosin binds: fluorescence resonance energy transfer detects an increase in the radial coordinate of Cys-374
Authors: Moens PD, dos Remedios CG.
Journal: Biochemistry (1997): 7353
Authors: Moens PD, dos Remedios CG.
Journal: Biochemistry (1997): 7353
Interhead distances in myosin attached to F-actin estimated by fluorescence energy transfer spectroscopy
Authors: Ishiwata S, Miki M, Shin I, Funatsu T, Yasuda K, dos Remedios CG.
Journal: Biophys J (1997): 895
Authors: Ishiwata S, Miki M, Shin I, Funatsu T, Yasuda K, dos Remedios CG.
Journal: Biophys J (1997): 895
Myosin-induced changes in F-actin: fluorescence probing of subdomain 2 by dansyl ethylenediamine attached to Gln-41
Authors: Kim E, Miller CJ, Motoki M, Seguro K, Muhlrad A, Reisler E.
Journal: Biophys J (1996): 1439
Authors: Kim E, Miller CJ, Motoki M, Seguro K, Muhlrad A, Reisler E.
Journal: Biophys J (1996): 1439
Changes in the distribution of F-actin in the fission yeast Schizosaccharomyces pombe by arresting growth in distilled water: correlative studies with fluorescence and electron microscopy
Authors: Kanbe T, Akashi T, Tanaka K.
Journal: J Electron Microsc (Tokyo) (1994): 20
Authors: Kanbe T, Akashi T, Tanaka K.
Journal: J Electron Microsc (Tokyo) (1994): 20
Application notes
A Novel Fluorescent Probe for Imaging and Detecting Hydroxyl Radical in Living Cells
Fluorescent Oligonucleotide Labeling Reagents
Monitoring of Mitochondrial Membrane Potential Changes in Live Cells Using JC-10
Selective Analysis of RNA in Live and Fixed Cells with StrandBrite RNA Green
Cell Loading Protocol For Fluorescent pH Indicator, BCECF-AM
Fluorescent Oligonucleotide Labeling Reagents
Monitoring of Mitochondrial Membrane Potential Changes in Live Cells Using JC-10
Selective Analysis of RNA in Live and Fixed Cells with StrandBrite RNA Green
Cell Loading Protocol For Fluorescent pH Indicator, BCECF-AM
FAQ
I ordered your phalloidin-amine (Cat #5302) so I can conjugate it to my oligo. Do you have a recommended protocol I can use?
What dye works best for staining and tracking lysosomes in live cells for several hours?
How can I lyse my cells without lysing the nuclear membrane?
Do you have any dual-fluorescence nucleic acid stains that interact with both DNA and RNA?
Do you have any fixable mitochondria staining assay kits?
What dye works best for staining and tracking lysosomes in live cells for several hours?
How can I lyse my cells without lysing the nuclear membrane?
Do you have any dual-fluorescence nucleic acid stains that interact with both DNA and RNA?
Do you have any fixable mitochondria staining assay kits?