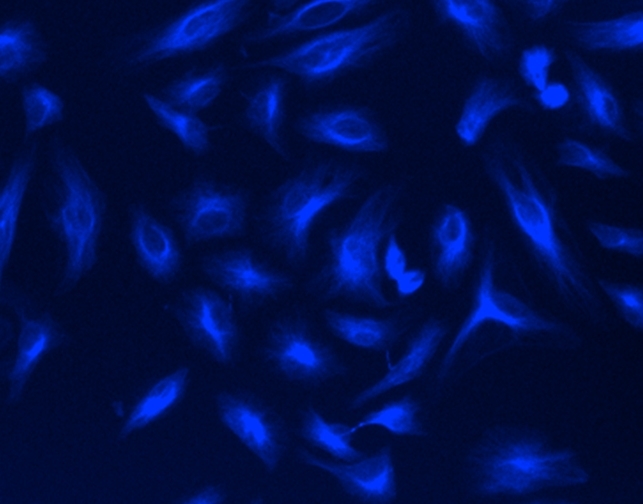
mFluor™ UV460 SE
AAT Bioquest's mFluor™ dyes are developed for multicolor flow cytometry-focused applications. These dyes have large Stokes Shifts, and can be well excited by the laser lines of flow cytometers (e.g., 355 nm, 405 nm, 488 nm and 633 nm). mFluor™ UV460 (MFUV460) dyes are well excited by UV excitation with emission at ~460 nm. MFUV460 has the spectral properties almost identical to those of Marina Blue® dye. These spectral characteristics make them an excellent alternative to Marina Blue® dye. Marina Blue® is the trademark of ThermoFisher.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 1136 | 1 mg | Price |
Physical properties
| Molecular weight | 367.26 |
| Solvent | DMSO |
Spectral properties
| Absorbance (nm) | 364 |
| Correction factor (260 nm) | 0.35 |
| Correction factor (280 nm) | 0.134 |
| Extinction coefficient (cm -1 M -1) | 15000 1 |
| Excitation (nm) | 358 |
| Emission (nm) | 456 |
| Quantum yield | 0.86 1 |
Storage, safety and handling
| Intended use | Research Use Only (RUO) |
| Storage | Freeze (< -15 °C); Minimize light exposure |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 6, 2026

