PhosphoWorks™ Fluorimetric Pyrophosphate Assay Kit
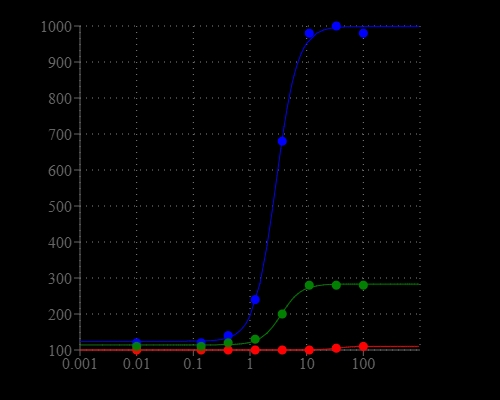
Blue Fluorescence
Pyrophosphate (PPi) is produced by a number of biochemical reactions, such as ATP hydrolysis, DNA and RNA polymerizations, cyclic AMP formation by the enzyme adenylate cyclase and the enzymatic activation of fatty acids to form their coenzyme A esters. Our PhosphoWroks™ Pyrophosphate Assay Kit provides the most robust spectrophotometric method for measuring pyrophosphate. This kit uses our proprietary fluorogenic pyrophosphate sensor that has its fluorescence intensity proportionally dependent upon the concentration of pyrophosphate. Our assay is much easier and more robust than the enzyme-coupling pyrophosphate methods that require at least two enzymes for their pyrophosphate detections. The kit provides all the essential components for assaying pyrophosphate. This kit has been successfully used in high throughput screening (HTS). Please inquire special HTS bulk package discount for the screening of >10,000 assays.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 21611 | 200 Tests | Price |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12352200 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 316 nm |
| Emission | 456 nm |
| Cutoff | 420 nm |
| Recommended plate | Solid black |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 4, 2026
