Calcein Red™ AM
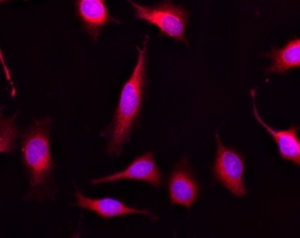
Calcein AM is one of the most popular fluorescent probes used for labeling and monitoring cellular functions of live cells. However, the single color of Calcein AM makes it impossible to use this valuable reagent in the multicolor applications. For example, it is impossible to use Calcein AM in combination of GFP-tranfacted cells due to the same color to GFP. To address this color limitation of Calcein AM, we have developed Calcein Orange™, Calcein Red™ and Calcein Deep Red™. These new Calcein AM analogs enable the multicolor labeling and functional analysis of live cells in combination with Calcein AM. Non-fluorescent Calcein Red™ AM can easily get into live cells and hydrolyzes to generate strongly fluorescent Calcein Red™ dye. Calcein Red™ dye can be monitored with the common TRITC/Cy3 filter set. AAT Bioquest offers Calcein Red™ as a reference dye to Calcein Red™ AM.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 21900 | 1 mg | Price |
Physical properties
| Molecular weight | 1015.79 |
| Solvent | DMSO |
Spectral properties
| Excitation (nm) | 562 |
| Emission (nm) | 576 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Storage | Freeze (< -15 °C); Minimize light exposure |
| UNSPSC | 12352200 |
Instrument settings
| Flow cytometer | |
| Excitation | 532/561 nm laser |
| Emission | 585/40 nm filter |
| Fluorescence microscope | |
| Excitation | TRITC filter set |
| Emission | TRITC filter set |
| Recommended plate | Black wall/clear bottom |
| Fluorescence microplate reader | |
| Excitation | 540 |
| Emission | 590 |
| Cutoff | 570 |
| Recommended plate | Black wall/clear bottom |
| Instrument specification(s) | Bottom read mode |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on January 23, 2025

