Amplite® Fluorimetric Glutathione Assay Kit
Green Fluorescence
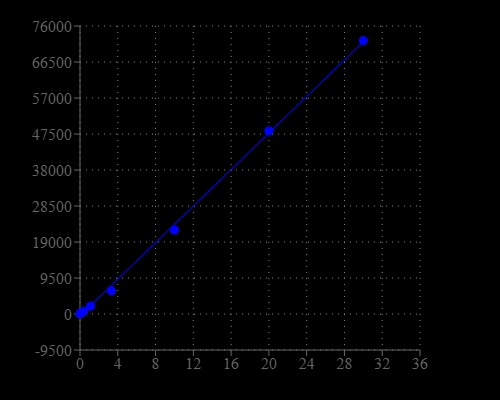
The monitoring of reduced and oxidized glutathione (GSH) in biological samples is essential for evaluating the redox and detoxification status of cells and tissues in relation to the protective role of glutathione against oxidative and free-radical-mediated cell injury. Cysteine metabolism disorders include cystinosis, an autosomal recessive disease produced by a defect in lysosomal transport, and cystinuria, a common heritable disorder of amino acid transport. Cysteine is unique among the amino acids found in proteins. There are a few reagents or assay kits available for quantitating thiols in biological systems. However, all the commercial kits either lack sensitivity or have tedious protocols. Our Amplite® Fluorimetric Glutathione Qutitation Kit provides an ultrasensitive fluorimetric assay to quantitate GSH in sample. The kit uses a proprietary non-fluorescent dye that becomes strongly fluorescent upon reacting with thiol. The kit provides a sensitive, one-step fluorimetric method to detect as little as 1 picomole of cysteine or GSH in a 100 µL assay volume. The assay can be performed in a convenient 96-well or 384-well microtiter-plate format and easily adapted to automation without a separation step. Its signal can be easily read using a fluorescence microplate reader.

| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 10055 | 200 Tests | Price |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12171501 |
Instrument settings
| Fluorescence microplate reader | |
| Excitation | 490 nm |
| Emission | 525 nm |
| Cutoff | 515 nm |
| Recommended plate | Solid black |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on May 11, 2026
