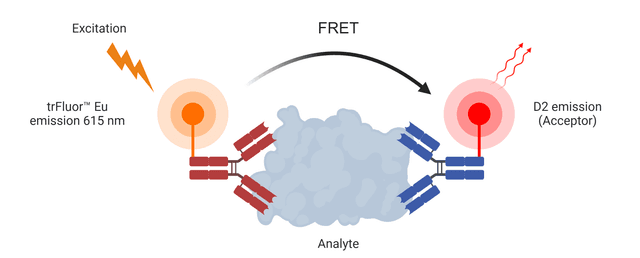
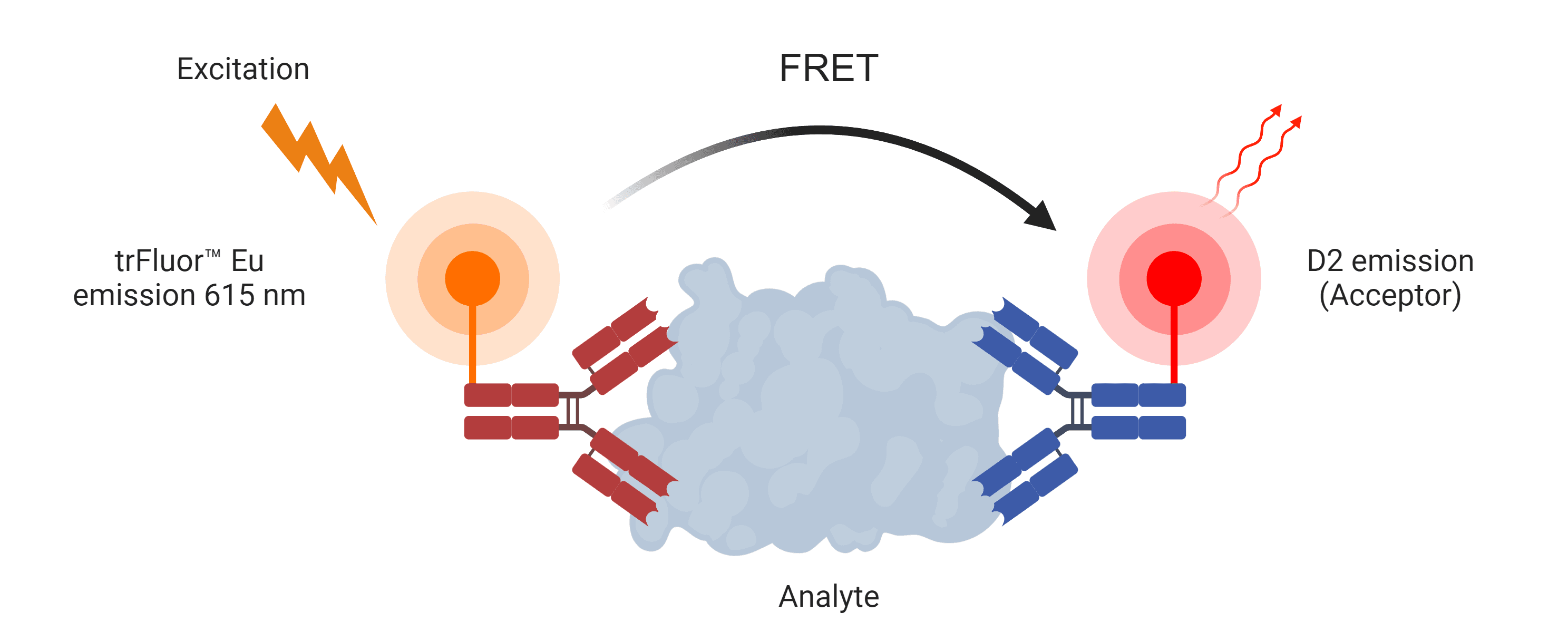
Buccutite™ Rapid trFluor™ D2 Acceptor Antibody Labeling Kit *Microscale Optimized for Labeling 100 ug Antibody Per Reaction*
| Price | |
| Catalog Number | |
| Unit Size | |
| Quantity |
| Telephone | 1-800-990-8053 |
| Fax | 1-800-609-2943 |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Shipping | Standard overnight for United States, inquire for international |
| Excitation (nm) | 651 |
| Emission (nm) | 660 |
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| Overview |
Excitation (nm) 651 | Emission (nm) 660 |
Components
Example protocol
AT A GLANCE
Add 5 µL Reaction Buffer (Component C) into the antibody solution (100 µL).
Add the antibody solution into Buccutite™ MTA vial (Component B).
Incubate at room temperature for 30 minutes.
Mix with 50 µL Buccutite™ FOL-Activated trFluor™ D2 working solution.
Incubate at room temperature for 60 minutes.
Please store the kit at 4°C upon receiving it. Ensure it is stored properly to maintain stability for six months. Alternatively, Component B can be stored at -20°C. Do not freeze Buccutite™ FOL-Activated trFluor™ D2 (Component A) or Reaction Buffer (Component C). Before opening the vials, warm all components and briefly centrifuge them. Prepare the required solutions immediately after opening the vials to begin your conjugation. For reference, an example SOP for labeling goat anti-mouse IgG antibodies is provided.
PREPARATION OF WORKING SOLUTION
For labeling 100 µg antibody (assuming the target antibody concentration is 1 mg/mL), mix 5 µL (5% of the total reaction volume) of Reaction Buffer (Component C) with 100 µL of the target antibody solution.
Note: If you have a different concentration, adjust the antibody volume accordingly to make ~100 µg antibody available for your labeling reaction.
Note: The antibody should be dissolved in 1X phosphate-buffered saline (PBS), pH 7.2-7.4; If the antibody is dissolved in glycine buffer, it must be dialyzed against 1X PBS, pH 7.2-7.4, or use ReadiUse™ 10KD Spin Filter (Cat. 60502 from AAT Bioquest) to remove free amines or ammonium salts (such as ammonium sulfate and ammonium acetate) that are widely used for antibody precipitation.
Note: Impure antibodies or antibodies stabilized with bovine serum albumin (BSA) or gelatin will not be labeled well.
Note: The antibody –Buccutite™ MTA reaction efficiency is significantly reduced if the antibody concentration is less than 1 mg/mL. For optimal labeling efficiency, the final antibody concentration range of 1-10 mg/mL is recommended.
SAMPLE EXPERIMENTAL PROTOCOL
Add the antibody working solution directly into the vial of Buccutite ™ MTA (Component B), and mix them well by repeatedly pipetting for a few times or vortex the vial for a few seconds.
Keep the antibody- Buccutite ™ MTA reaction mixture at room temperature for 30 - 60 minutes.
Note: The antibody-Buccutite™ MTA reaction mixture can be rotated or shaken for a longer time if desired.
Make Buccutite™ FOL-Activated trFluor™ D2 solution by adding 50 µL ddH2O into the vial of Buccutite™ FOL-Activated trFluor™ D2 (Component A), mix well by repeatedly pipetting for a few times or vortex the vial for a few seconds.
Mix the whole vial of Buccutite™ FOL-Activated trFluor™ D2 solution into the antibody-Buccutite™ MTA solution, mix well, and rotate the mixture for 1 hour at room temperature.
The antibody-trFluor™ D2 conjugate is now ready to use.
Note: For immediate use, the antibody-trFluor™ D2 conjugate needs to be diluted with the buffer of your choice.
The antibody conjugate should be stored at > 0.5 mg/mL in the presence of a carrier protein (e.g., 0.1% bovine serum albumin). The Antibody-trFluor™ D2 conjugate solution could be stored at 4 °C for two months without significant change when stored in the presence of 2 mM sodium azide and kept from light. For longer storage, the antibody-trFluor™ D2 conjugates could be lyophilized and stored at ≤ –20 °C.
Images
References
Authors: Larson, Jacob E and Hardy, P Brian and Schomburg, Noah K and Wang, Xiaodong and Kireev, Dmitri and Rossman, Kent L and Pearce, Kenneth H
Journal: SLAS discovery : advancing life sciences R & D (2023): 39-47
Authors: Roth, Doris and Benz, Jörg and Grether, Uwe and Dietz, Michel
Journal: Methods in molecular biology (Clifton, N.J.) (2023): 155-169
Authors: Iqbal, Saleem and Lin, Sheng-Xiang
Journal: Bioengineering (Basel, Switzerland) (2023)
Authors: Bux, Jessica and Sen, Nesli Ece and Klink, Isa-Maria and Hauser, Stefan and Synofzik, Matthis and Schöls, Ludger and Auburger, Georg and Riess, Olaf and Hübener-Schmid, Jeannette
Journal: Molecular neurobiology (2023): 3553-3567
Authors: Abdel-Rahman, Somaya A and Zhang, Longfei and Gabr, Moustafa T
Journal: SLAS discovery : advancing life sciences R & D (2023): 188-192
Authors: Yue, Hong and Nowak, Radosław P and Overwijn, Daan and Payne, N Connor and Fischinger, Stephanie and Atyeo, Caroline and Lam, Evan C and St Denis, Kerri and Brais, Lauren K and Konishi, Yoshinobu and Sklavenitis-Pistofidis, Romanos and Baden, Lindsey R and Nilles, Eric J and Karlson, Elizabeth W and Yu, Xu G and Li, Jonathan Z and Woolley, Ann E and Ghobrial, Irene M and Meyerhardt, Jeffrey A and Balazs, Alejandro B and Alter, Galit and Mazitschek, Ralph and Fischer, Eric S
Journal: Cell reports methods (2023): 100421
Authors: Cecon, Erika and Dam, Julie and Jockers, Ralf
Journal: STAR protocols (2022): 101024
Authors: Gorshkov, Kirill and Morales Vasquez, Desarey and Chiem, Kevin and Ye, Chengjin and Nguyen Tran, Bruce and Carlos de la Torre, Juan and Moran, Thomas and Chen, Catherine Z and Martinez-Sobrido, Luis and Zheng, Wei
Journal: ACS pharmacology & translational science (2022): 8-19
Authors: Chen, Heng and Feng, Yongtong and Cao, Yang and Tang, Yuguo and Liu, Tao
Journal: The Analyst (2022): 318-324
Authors: Cecon, Erika and Burridge, Matilda and Cao, Longxing and Carter, Lauren and Ravichandran, Rashmi and Dam, Julie and Jockers, Ralf
Journal: Cell chemical biology (2022): 74-83.e4
Application notes
A Novel Fluorescent Probe for Imaging and Detecting Hydroxyl Radical in Living Cells
A Novel NO Wash Probeniceid-Free Calcium Assay for Functional Analysis of GPCR and Calcium Channel Targets
Biotin Labeling Molecules and Their Biological Applications
Buccutite™ Bioconjugation Technology
FAQ
What are the differences between calcium ion indicators: Cal 520, Cal 520FF, and Cal 520N?
How do I make an AM ester stock solution?
Can we fix cells with glutaraldehyde and then stain with fluorescent phalloidin?
What is the difference between FluoroQuest Anti-fading Kit I and FluoroQuest Anti-fading Kit II?