ReadiLink™ Rapid iFluor® 594 Antibody Labeling Kit
Microscale Optimized for Labeling 50 μg Antibody Per Reaction
AAT Bioquest's iFluor® dyes are optimized for labeling proteins, in particular, antibodies. iFluor® 594 dyes have fluorescence excitation and emission maxima of ~590 nm and ~610 nm respectively. iFluor® 594 family has the spectral properties similar to those of Texas Red® and Alexa Fluor® 594 (Texas Red® and Alexa Fluor® 594 are the trademarks of Invitrogen). iFluor® 594 family is pH-independent from pH 3 to 11. These spectral characteristics make this new dye family an excellent alternative to Texas Red® and Alexa Fluor® 594.Compared to Texas Red®, iFluor® 594 is much easier to be conjugated with RPE with much higher conjugation yield, and the resulted RPE-iFluor® 594 tandem has better FRET efficiency.ReadiLink™ labeling kits essentially only require 2 simple mixing steps without a column purification needed. iFluor® 594 SE used in this ReadiLink™ kit is reasonably stable and shows good reactivity and selectivity with protein amino groups. The kit has all the essential components for labeling ~2x50 ug antibody. Each of the two vials of iFluor® 594 dye provided in the kit is optimized for labeling ~50 µg antibody. iFluor® 594 SE protein labeling kit provides a convenient method to label monoclonal, polyclonal antibodies or other proteins (>10 kDa) with the iFluor® 594 SE.
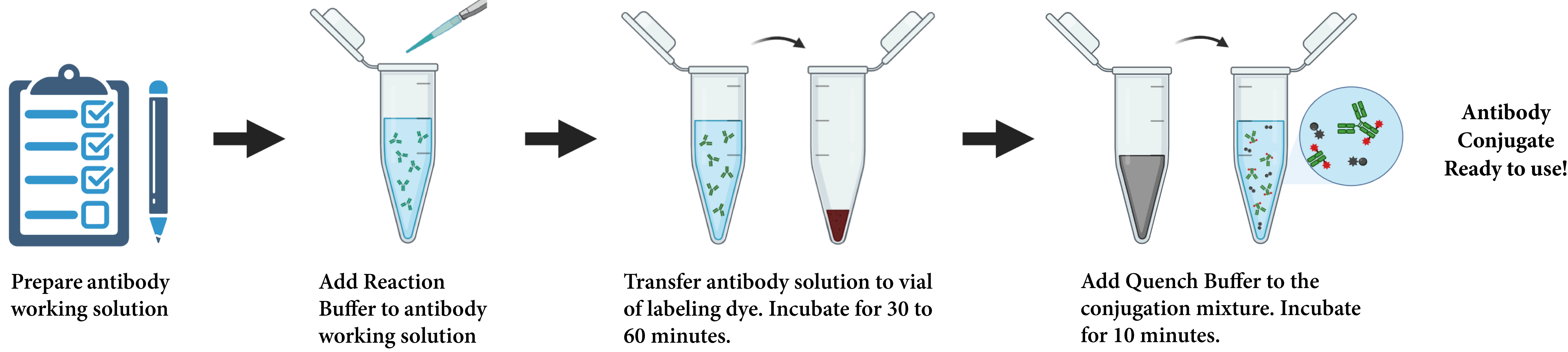
Figure 1. Overview of the ReadiLink™ Rapid Antibody Labeling protocol. In just two simple steps, and with no purification necessary, covalently label microgram amounts of antibodies in under an hour.


| Catalog | Size | Price | Quantity |
|---|---|---|---|
| 1230 | 2 Labelings | Price |
Spectral properties
| Absorbance (nm) | 587 |
| Correction factor (260 nm) | 0.05 |
| Correction factor (280 nm) | 0.04 |
| Extinction coefficient (cm -1 M -1) | 200000 1 |
| Excitation (nm) | 587 |
| Emission (nm) | 603 |
| Quantum yield | 0.53 1 |
Storage, safety and handling
| H-phrase | H303, H313, H333 |
| Hazard symbol | XN |
| Intended use | Research Use Only (RUO) |
| R-phrase | R20, R21, R22 |
| UNSPSC | 12171501 |
Contact us
| Telephone | |
| Fax | |
| sales@aatbio.com | |
| International | See distributors |
| Bulk request | Inquire |
| Custom size | Inquire |
| Technical Support | Contact us |
| Request quotation | Request |
| Purchase order | Send to sales@aatbio.com |
| Shipping | Standard overnight for United States, inquire for international |
Page updated on June 5, 2026

