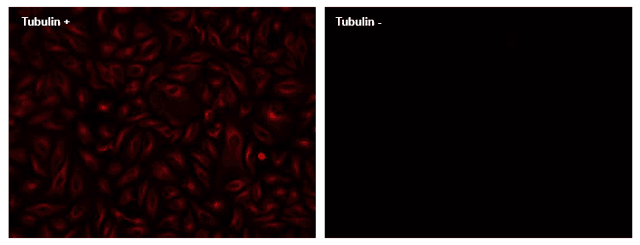
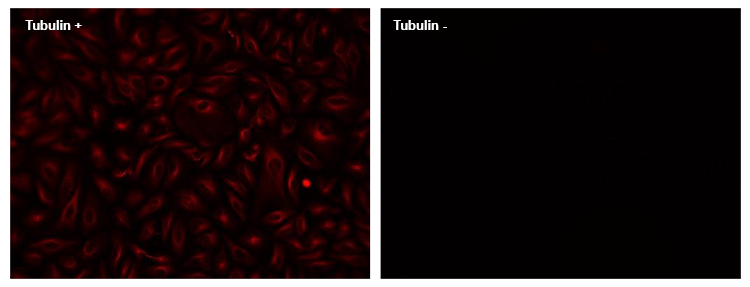
ReadiLink™ Rapid XFD750 Antibody Labeling Kit *XFD750 Same Structure to Alexa Fluor™ 750*
Example protocol
AT A GLANCE
Warm all the components and centrifuge the vials briefly before opening them. Immediately prepare the necessary solutions before starting your conjugation. The following protocol is a recommendation.
PREPARATION OF WORKING SOLUTION
For labeling 50 µg of protein (assuming the target protein concentration is 1 mg/mL), mix 5 µL (10% of the total reaction volume) of Reaction Buffer (Component B) with 50 µL of the target protein solution.
Note: If you have a different protein concentration, adjust the protein volume accordingly to make ~50 µg of protein available for your labeling reaction.
Note: For labeling 100 µg of protein (assuming the target protein concentration is 1 mg/mL), mix 10 µL (10% of the total reaction volume) of Reaction Buffer (Component B) with 100 µL of the target protein solution.
Note: The protein should be dissolved in 1X phosphate buffered saline (PBS), pH 7.2 - 7.4; if the protein is dissolved in glycine buffer, it must be dialyzed against 1X PBS, pH 7.2 - 7.4, or use Amicon Ultra-0.5, Ultracel-10 Membrane, 10 kDa (Cat. UFC501008 from Millipore) to remove free amines or ammonium salts (such as ammonium sulfate and ammonium acetate) that are widely used for protein precipitation.
Note: Impure antibodies or antibodies stabilized with bovine serum albumin (BSA) or gelatin will not be labeled well.
Note: A final protein concentration range of 1-2 mg/mL is recommended for optimal labeling efficiency, with a significantly reduced conjugation efficiency at less than 1 mg/mL.
SAMPLE EXPERIMENTAL PROTOCOL
Add the protein working solution (Solution A) to ONE vial of labeling dye (Component A), and mix them well by repeatedly pipetting for a few times or vortex the vial for a few seconds.
Note: If labeling 100 µg of protein, use both vials (Component A) of labeling dye by dividing the 100 µg of protein into 2 x 50 µg of protein and reacting each 50 µg of protein with one vial of labeling dye. Then combine both vials for the next step.
Keep the conjugation reaction mixture at room temperature for 30-60 minutes.
Note: The conjugation reaction mixture can be rotated or shaken for a longer time if desired.
Add 5 µL (for 50 µg protein) or 10 µL (for 100 µg protein) which is 10% of the total reaction volume of TQ™-Dyed Quench Buffer (Component C) into the conjugation reaction mixture; mix well.
Incubate at room temperature for 10 minutes. The labeled protein (antibody) is now ready to use.
The protein conjugate should be stored at > 0.5 mg/mL in the presence of a carrier protein (e.g., 0.1% bovine serum albumin). For longer storage, the protein conjugates could be lyophilized or divided into single-used aliquots and stored at ≤ –20°C.
Spectrum
Product family
References
Authors: Fuenzalida Werner, Juan Pablo and Huang, Yuanhui and Mishra, Kanuj and Janowski, Robert and Vetschera, Paul and Heichler, Christina and Chmyrov, Andriy and Neufert, Clemens and Niessing, Dierk and Ntziachristos, Vasilis and Stiel, Andre C
Journal: Analytical chemistry (2020)
Authors: Kleinmanns, Katrin and Fosse, Vibeke and Davidson, Ben and de Jalón, Elvira García and Tenstad, Olav and Bjørge, Line and McCormack, Emmet
Journal: EBioMedicine (2020): 102783
Authors: Duranti, Claudia and Carraresi, Laura and Sette, Angelica and Stefanini, Matteo and Lottini, Tiziano and Crescioli, Silvia and Crociani, Olivia and Iamele, Luisa and De Jonge, Hugo and Gherardi, Ermanno and Arcangeli, Annarosa
Journal: Oncotarget (2018): 34972-34989
Authors: Rastogi, Shiva K and Anderson, Hailee E and Lamas, Joseph and Barret, Scott and Cantu, Travis and Zauscher, Stefan and Brittain, William J and Betancourt, Tania
Journal: ACS applied materials & interfaces (2018): 30071-30080
Authors: Dong, Lili and Zhou, Hongxin and Zhao, Menglong and Gao, Xinghui and Liu, Yang and Liu, Dongli and Guo, Wei and Hu, Hongwei and Xie, Qian and Fan, Jia and Lin, Jiang and Wu, Weizhong
Journal: Molecular therapy. Nucleic acids (2018): 376-386