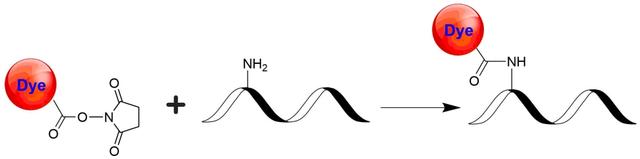
iFluor® 840 succinimidyl ester
In vivo fluorescence imaging uses a sensitive camera to detect the fluorescence emission from fluorophores in whole-body living small animals. To overcome the photon attenuation in living tissue, fluorophores with long emission at the infrared (IR) region are generally preferred. Recent advances in imaging strategies and reporter techniques for in vivo fluorescence imaging include novel approaches to improve the specificity and affinity of the probes and to modulate and amplify the signal at target sites for enhanced sensitivity. Further emerging developments aim to achieve high-resolution, multimodality, and lifetime-based in vivo fluorescence imaging. Our iFluor® 840 is designed to label proteins and other biomolecules with infrared fluorescence. Conjugates prepared with iFluor® 840 have excitation and emission in the IR range. iFluor® 840 dye emission is well separated from commonly used far-red fluorophores such as Cy5, Cy7, or allophycocyanin (APC), facilitating multicolor analysis. This fluorophore is also useful for small animal in vivo imaging applications or other imaging applications requiring IR detection. iFluor® 840 succinimidyl ester is amine-reactive and can be readily used to conjugate amine-containing biomolecules, particularly antibodies.
Example protocol
PREPARATION OF STOCK SOLUTIONS
Unless otherwise noted, all unused stock solutions should be divided into single-use aliquots and stored at -20 °C after preparation. Avoid repeated freeze-thaw cycles.
Note The pH of the protein solution (Solution A) should be 8.5 ± 0.5. If the pH of the protein solution is lower than 8.0, adjust the pH to the range of 8.0-9.0 using 1 M sodium bicarbonate solution or 1 M pH 9.0 phosphate buffer.
Note The protein should be dissolved in 1X phosphate buffered saline (PBS), pH 7.2-7.4. If the protein is dissolved in Tris or glycine buffer, it must be dialyzed against 1X PBS, pH 7.2-7.4, to remove free amines or ammonium salts (such as ammonium sulfate and ammonium acetate) that are widely used for protein precipitation.
Note Impure antibodies or antibodies stabilized with bovine serum albumin (BSA) or gelatin will not be labeled well. The presence of sodium azide or thimerosal might also interfere with the conjugation reaction. Sodium azide or thimerosal can be removed by dialysis or spin column for optimal labeling results.
Note The conjugation efficiency is significantly reduced if the protein concentration is less than 2 mg/mL. For optimal labeling efficiency the final protein concentration range of 2-10 mg/mL is recommended.
Note Prepare the dye stock solution (Solution B) before starting the conjugation. Use promptly. Extended storage of the dye stock solution may reduce the dye activity. Solution B can be stored in freezer for two weeks when kept from light and moisture. Avoid freeze-thaw cycles.
1. Protein stock solution (Solution A)
Mix 100 µL of a reaction buffer (e.g., 1 M sodium carbonate solution or 1 M phosphate buffer with pH ~9.0) with 900 µL of the target protein solution (e.g. antibody, protein concentration >2 mg/mL if possible) to give 1 mL protein labeling stock solution.Note The pH of the protein solution (Solution A) should be 8.5 ± 0.5. If the pH of the protein solution is lower than 8.0, adjust the pH to the range of 8.0-9.0 using 1 M sodium bicarbonate solution or 1 M pH 9.0 phosphate buffer.
Note The protein should be dissolved in 1X phosphate buffered saline (PBS), pH 7.2-7.4. If the protein is dissolved in Tris or glycine buffer, it must be dialyzed against 1X PBS, pH 7.2-7.4, to remove free amines or ammonium salts (such as ammonium sulfate and ammonium acetate) that are widely used for protein precipitation.
Note Impure antibodies or antibodies stabilized with bovine serum albumin (BSA) or gelatin will not be labeled well. The presence of sodium azide or thimerosal might also interfere with the conjugation reaction. Sodium azide or thimerosal can be removed by dialysis or spin column for optimal labeling results.
Note The conjugation efficiency is significantly reduced if the protein concentration is less than 2 mg/mL. For optimal labeling efficiency the final protein concentration range of 2-10 mg/mL is recommended.
2. iFluor™ 840 SE stock solution (Solution B)
Add anhydrous DMSO into the vial of iFluor™ 840 SE to make a 10 mM stock solution. Mix well by pipetting or vortex.Note Prepare the dye stock solution (Solution B) before starting the conjugation. Use promptly. Extended storage of the dye stock solution may reduce the dye activity. Solution B can be stored in freezer for two weeks when kept from light and moisture. Avoid freeze-thaw cycles.
SAMPLE EXPERIMENTAL PROTOCOL
This labeling protocol was developed for the conjugate of Goat anti-mouse IgG with iFluor™ 840 SE. You might need further optimization for your particular proteins.
Note Each protein requires distinct dye/protein ratio, which also depends on the properties of dyes. Over labeling of a protein could detrimentally affects its binding affinity while the protein conjugates of low dye/protein ratio gives reduced sensitivity.
Note Each protein requires distinct dye/protein ratio, which also depends on the properties of dyes. Over labeling of a protein could detrimentally affects its binding affinity while the protein conjugates of low dye/protein ratio gives reduced sensitivity.
Run conjugation reaction
- Use 10:1 molar ratio of Solution B (dye)/Solution A (protein) as the starting point: Add 5 µL of the dye stock solution (Solution B, assuming the dye stock solution is 10 mM) into the vial of the protein solution (95 µL of Solution A) with effective shaking. The concentration of the protein is ~0.05 mM assuming the protein concentration is 10 mg/mL and the molecular weight of the protein is ~200KD.
Note We recommend to use 10:1 molar ratio of Solution B (dye)/Solution A (protein). If it is too less or too high, determine the optimal dye/protein ratio at 5:1, 15:1 and 20:1 respectively. - Continue to rotate or shake the reaction mixture at room temperature for 30-60 minutes.
Purify the conjugation
The following protocol is an example of dye-protein conjugate purification by using a Sephadex G-25 column.- Prepare Sephadex G-25 column according to the manufacture instruction.
- Load the reaction mixture (From "Run conjugation reaction") to the top of the Sephadex G-25 column.
- Add PBS (pH 7.2-7.4) as soon as the sample runs just below the top resin surface.
- Add more PBS (pH 7.2-7.4) to the desired sample to complete the column purification. Combine the fractions that contain the desired dye-protein conjugate.
Note For immediate use, the dye-protein conjugate need be diluted with staining buffer, and aliquoted for multiple uses.
Note For longer term storage, dye-protein conjugate solution need be concentrated or freeze dried.
Calculators
Common stock solution preparation
Table 1. Volume of DMSO needed to reconstitute specific mass of iFluor® 840 succinimidyl ester to given concentration. Note that volume is only for preparing stock solution. Refer to sample experimental protocol for appropriate experimental/physiological buffers.
| 0.1 mg | 0.5 mg | 1 mg | 5 mg | 10 mg | |
| 1 mM | 70.323 µL | 351.617 µL | 703.235 µL | 3.516 mL | 7.032 mL |
| 5 mM | 14.065 µL | 70.323 µL | 140.647 µL | 703.235 µL | 1.406 mL |
| 10 mM | 7.032 µL | 35.162 µL | 70.323 µL | 351.617 µL | 703.235 µL |
Molarity calculator
Enter any two values (mass, volume, concentration) to calculate the third.
| Mass (Calculate) | Molecular weight | Volume (Calculate) | Concentration (Calculate) | Moles | ||||
| / | = | x | = |
Spectrum
Product family
| Name | Excitation (nm) | Emission (nm) | Extinction coefficient (cm -1 M -1) | Quantum yield | Correction Factor (260 nm) | Correction Factor (280 nm) |
| iFluor® 350 succinimidyl ester | 345 | 450 | 200001 | 0.951 | 0.83 | 0.23 |
| iFluor® 405 succinimidyl ester | 403 | 427 | 370001 | 0.911 | 0.48 | 0.77 |
| iFluor® 488 succinimidyl ester | 491 | 516 | 750001 | 0.91 | 0.21 | 0.11 |
| iFluor® 514 succinimidyl ester | 511 | 527 | 750001 | 0.831 | 0.265 | 0.116 |
| iFluor® 532 succinimidyl ester | 537 | 560 | 900001 | 0.681 | 0.26 | 0.16 |
| iFluor® 555 succinimidyl ester | 557 | 570 | 1000001 | 0.641 | 0.23 | 0.14 |
| iFluor® 594 succinimidyl ester | 587 | 603 | 2000001 | 0.531 | 0.05 | 0.04 |
| iFluor® 633 succinimidyl ester | 640 | 654 | 2500001 | 0.291 | 0.062 | 0.044 |
| iFluor® 647 succinimidyl ester | 656 | 670 | 2500001 | 0.251 | 0.03 | 0.03 |
Show More (36) | ||||||
Citations
View all 8 citations: Citation Explorer
Block Face Scanning Electron Microscopy of Fluorescently Labeled Axons Without Using Near Infra-Red Branding
Authors: Maclachlan, C., Sahlender, D. A., Hayashi, S., Molnar, Z., Knott, G.
Journal: Front Neuroanat (2018): 88
Authors: Maclachlan, C., Sahlender, D. A., Hayashi, S., Molnar, Z., Knott, G.
Journal: Front Neuroanat (2018): 88
Bioactive magnetic near Infra-Red fluorescent core-shell iron oxide/human serum albumin nanoparticles for controlled release of growth factors for augmentation of human mesenchymal stem cell growth and differentiation
Authors: Levy, I., Sher, I., Corem-Salkmon, E., Ziv-Polat, O., Meir, A., Treves, A. J., Nagler, A., Kalter-Leibovici, O., Margel, S., Rotenstreich, Y.
Journal: J Nanobiotechnology (2015): 34
Authors: Levy, I., Sher, I., Corem-Salkmon, E., Ziv-Polat, O., Meir, A., Treves, A. J., Nagler, A., Kalter-Leibovici, O., Margel, S., Rotenstreich, Y.
Journal: J Nanobiotechnology (2015): 34
Novel near infra-red fluorescent pH sensors based on 1-aminoperylene bisimides covalently grafted onto poly(acryloylmorpholine)
Authors: Aigner, D., Borisov, S. M., Petritsch, P., Klimant, I.
Journal: Chem Commun (Camb) (2013): 2139-41
Authors: Aigner, D., Borisov, S. M., Petritsch, P., Klimant, I.
Journal: Chem Commun (Camb) (2013): 2139-41
In vitro and ex vivo evaluation of smart infra-red fluorescent caspase-3 probes for molecular imaging of cardiovascular apoptosis
Authors: Debunne, M., Portal, C., Delest, B., Brakenhielm, E., Lallem and , F., Henry, J. P., Ligeret, H., Noack, P., Massonneau, M., Romieu, A., Renard, P. Y., Thuillez, C., Richard, V.
Journal: Int J Mol Imaging (2011): 413290
Authors: Debunne, M., Portal, C., Delest, B., Brakenhielm, E., Lallem and , F., Henry, J. P., Ligeret, H., Noack, P., Massonneau, M., Romieu, A., Renard, P. Y., Thuillez, C., Richard, V.
Journal: Int J Mol Imaging (2011): 413290
Simple sensitive and simultaneous high-performance liquid chromatography method of glucoconjugated and non-glucoconjugated porphyrins and chlorins using near infra-red fluorescence detection
Authors: Canada-Canada, F., Bautista-Sanchez, A., Taverna, M., Prognon, P., Maillard, P., Grierson, D. S., Kasselouri, A.
Journal: J Chromatogr B Analyt Technol Biomed Life Sci (2005): 166-72
Authors: Canada-Canada, F., Bautista-Sanchez, A., Taverna, M., Prognon, P., Maillard, P., Grierson, D. S., Kasselouri, A.
Journal: J Chromatogr B Analyt Technol Biomed Life Sci (2005): 166-72
References
View all 19 references: Citation Explorer
Nanovesicle delivery to the liver via retinol binding protein and platelet-derived growth factor receptors: how targeting ligands affect biodistribution
Authors: Hsu, Ching-Yun and Chen, Chun-Han and Aljuffali, Ibrahim A and Dai, You-Shan and Fang, Jia-You
Journal: Nanomedicine (2017)
Authors: Hsu, Ching-Yun and Chen, Chun-Han and Aljuffali, Ibrahim A and Dai, You-Shan and Fang, Jia-You
Journal: Nanomedicine (2017)
A protocol for imaging alternative splicing regulation in vivo using fluorescence reporters in transgenic mice
Authors: Bonano VI, Oltean S, Garcia-Blanco MA.
Journal: Nat Protoc (2007): 2166
Authors: Bonano VI, Oltean S, Garcia-Blanco MA.
Journal: Nat Protoc (2007): 2166
A target cell-specific activatable fluorescence probe for in vivo molecular imaging of cancer based on a self-quenched avidin-rhodamine conjugate
Authors: Hama Y, Urano Y, Koyama Y, Kamiya M, Bernardo M, Paik RS, Shin IS, Paik CH, Choyke PL, Kobayashi H.
Journal: Cancer Res (2007): 2791
Authors: Hama Y, Urano Y, Koyama Y, Kamiya M, Bernardo M, Paik RS, Shin IS, Paik CH, Choyke PL, Kobayashi H.
Journal: Cancer Res (2007): 2791
Fluorescence imaging in vivo: recent advances
Authors: Rao J, Dragulescu-Andrasi A, Yao H.
Journal: Curr Opin Biotechnol (2007): 17
Authors: Rao J, Dragulescu-Andrasi A, Yao H.
Journal: Curr Opin Biotechnol (2007): 17
Ex vivo fluorescence imaging of normal and malignant urothelial cells to enhance early diagnosis
Authors: Steenkeste K, Lecart S, Deniset A, Pernot P, Eschwege P, Ferlicot S, Leveque-Fort S, Bri and et R, Fontaine-Aupart MP.
Journal: Photochem Photobiol (2007): 1157
Authors: Steenkeste K, Lecart S, Deniset A, Pernot P, Eschwege P, Ferlicot S, Leveque-Fort S, Bri and et R, Fontaine-Aupart MP.
Journal: Photochem Photobiol (2007): 1157
Page updated on September 19, 2024